Diethyl chlorophosphate
Diethylphosphoryl chloride ; Diethyl chlorophosphonate ; O,O-Diethyl phosphorochloridate ; Phosphoric acid diethyl ester chloride ; Phosphorochloridic acid O,O-diethyl ester ; Diethyl phosphorochloridate
For more information or to place an inquiry, please email us to
georganics@georganics.sk or use our contact form
Regulatory Information

H300 – Fatal if swallowed.
H310 – Fatal in contact with skin.
H331 – Toxic if inhaled.
P262 – Do not get in eyes, on skin, or on clothing.
P264 – Wash … thoroughly after handling.
P280 – Wear protective gloves/protective clothing/eye protection/face protection.
P301+P310 – IF SWALLOWED: Immediately call a POISON CENTER or doctor/physician.
P302+P352+P310 – IF ON SKIN: Wash with plenty of water. Immediately call a POISON CENTER/ doctor.
P304+P340 – IF INHALED: Remove victim to fresh air and keep at rest in a position comfortable for breathing.
P311 – Call a POISON CENTER or doctor/physician.
Product categorization
Description
By using Diethyl chlorophosphate, some ketones can be converted to enol phosphates which can be reduced to alkenes/alkanes or coupled with…
Show full descriptionGeneral description of Diethyl chlorophosphate:
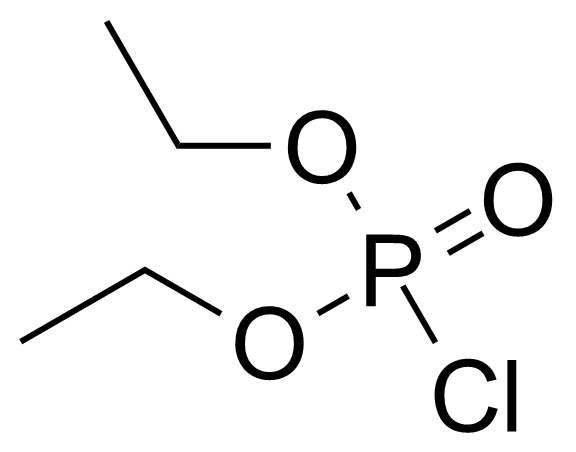
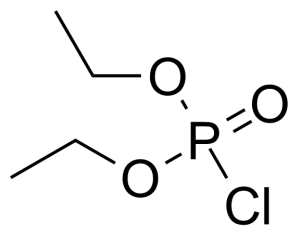 Diethyl chlorophosphate [814-49-3] or diethyl phosphorochloridate is a colorless to faith yellow clear liquid with the fruity odor and the boiling point of 60 °C/2 mmHg.[1] It acts as an cholinesterase inhibitor. It has high oral (LD50 = 11 mg/kg, rat) and very high dermal toxicity (LD50 = 8 μL/kg, rabbit), it is also toxic by inhalation.[2] This compound is typically prepared by the chlorination of diethylphosphite with carbon tetrachloride, which is called Atherton–Todd reaction.[3] Another option of preparation is reaction of phosphoryl chloride with ethanol in the presence of triethylamine.[4]
Diethyl chlorophosphate [814-49-3] or diethyl phosphorochloridate is a colorless to faith yellow clear liquid with the fruity odor and the boiling point of 60 °C/2 mmHg.[1] It acts as an cholinesterase inhibitor. It has high oral (LD50 = 11 mg/kg, rat) and very high dermal toxicity (LD50 = 8 μL/kg, rabbit), it is also toxic by inhalation.[2] This compound is typically prepared by the chlorination of diethylphosphite with carbon tetrachloride, which is called Atherton–Todd reaction.[3] Another option of preparation is reaction of phosphoryl chloride with ethanol in the presence of triethylamine.[4]Application of Diethyl chlorophosphate:
By using this substance, some ketones can be converted to enol phosphates which can be reduced to alkenes/alkanes or coupled with organometallic reagents to form substituted alkenes. Following enol phosphates can be then converted into β-keto phosphonates, useful for Horner-Emmons homologation.[5] It is also used in the synthesis of organophosphorus nerve agent mimics.[6] Phosphoroamidate linkages are found in a large array of biologically active natural products for example Microcin C7, Dinogunellin, Phosphoarginine, Phosphocreatine, Phosphoramidon, Phosmidosine and Agrocin.[7]Product categorization (Chemical groups):
Main category: ______________________________________________________________________________________Similar products
| Product name | Structure | CAS# | G-code | |
|---|---|---|---|---|
| 2-Aminoethyl dihydrogen phosphate | 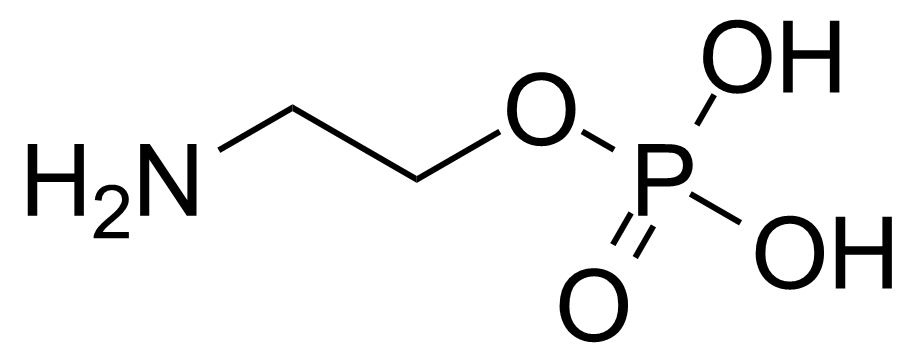 | [1071-23-4] | GEO-00129 | |
| 2-Aminoethyl dihydrogen phosphate magnesium salt (2:1) | 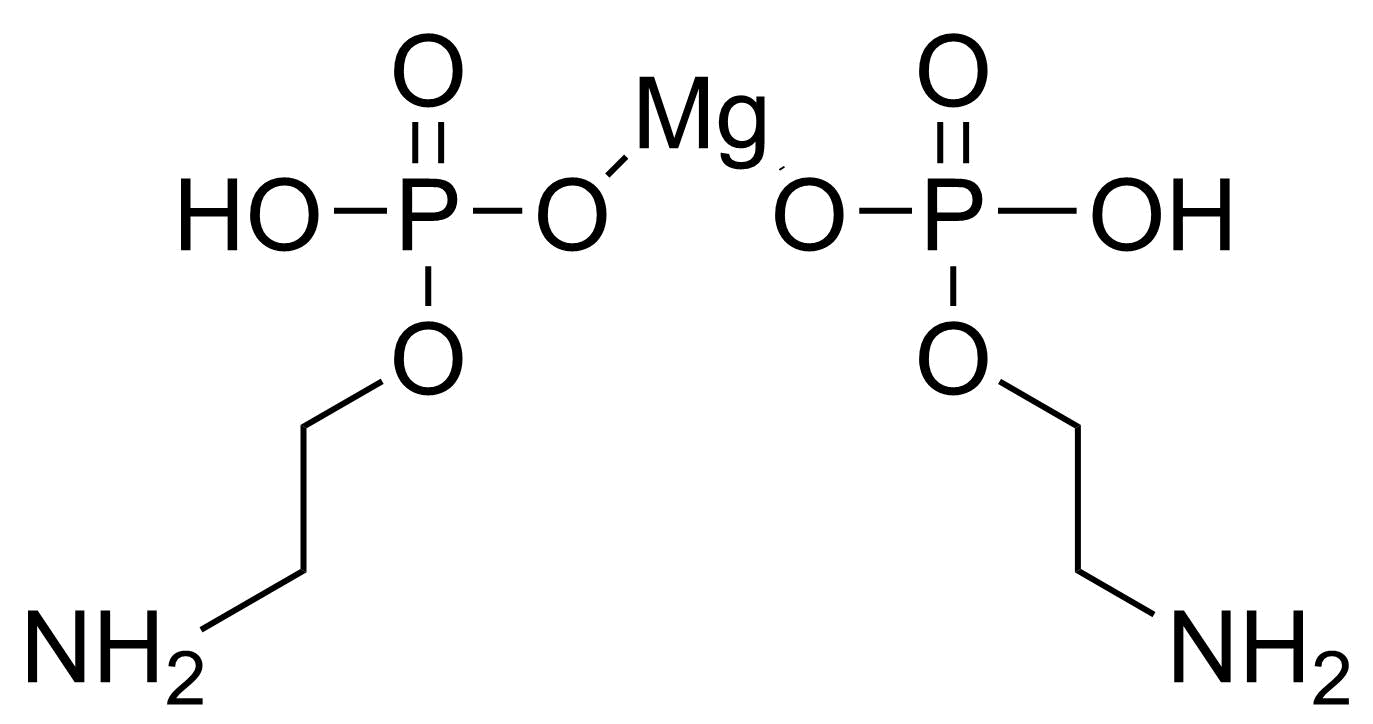 | N/A | GEO-04286 | |
| (Aminomethyl)phosphonic acid | 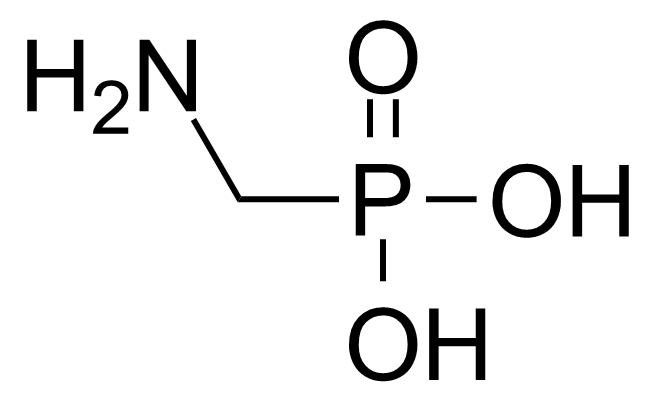 | [1066-51-9] | GEO-00169 | |
| (3-Aminopropyl)phosphonic acid | 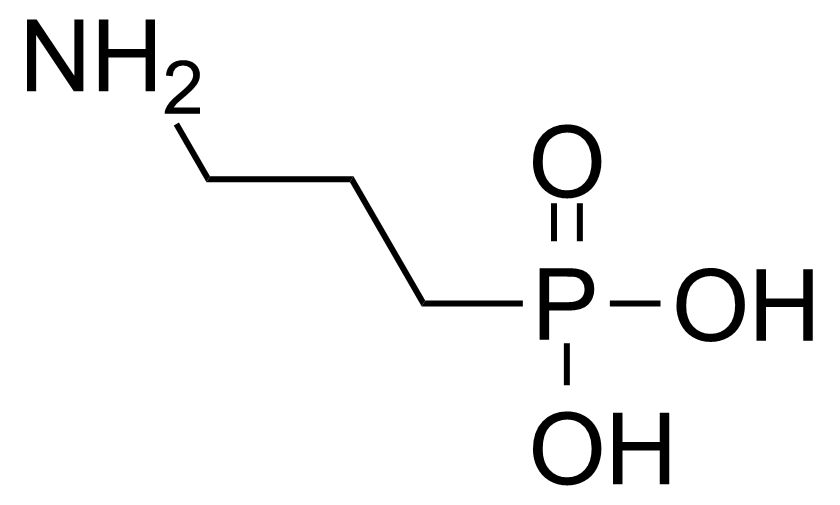 | [13138-33-5] | GEO-00218 | |
| Benzo[d]isoxazol-3-yl diphenyl phosphate | ![Structure of Benzo[d]isoxazol-3-yl diphenyl phosphate](https://georganics.sk/wp-content/uploads/2021/05/GEO-00266_Benzodisoxazol-3-yl_diphenyl_phosphate.png) | [94820-31-2] | GEO-00266 | |
| Benzyl dimethylphosphonoacetate | 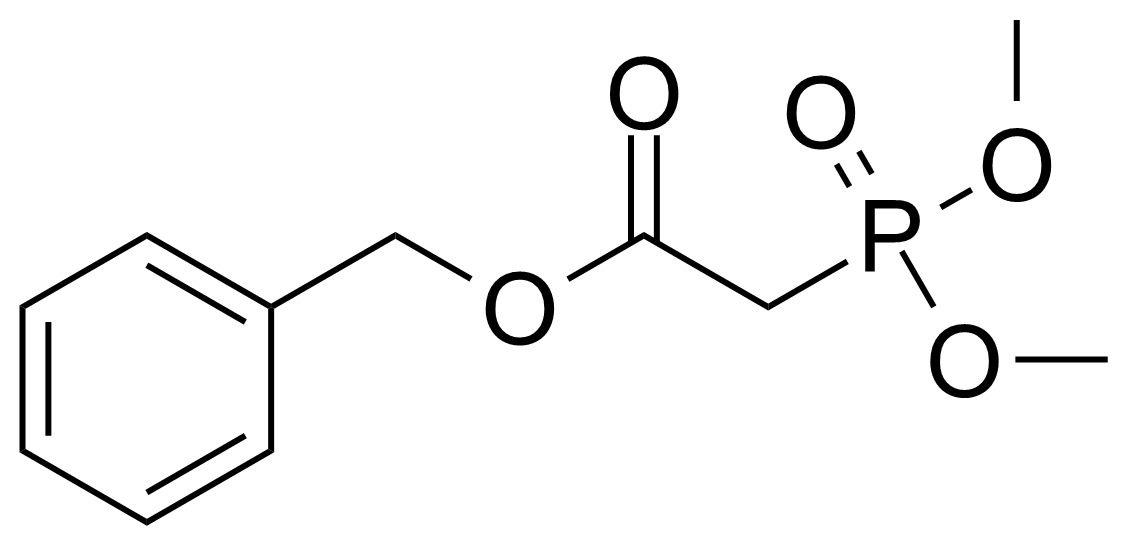 | [57443-18-2] | GEO-02882 | |
| Benzyl(triphenylphosphoranylidene)acetate | 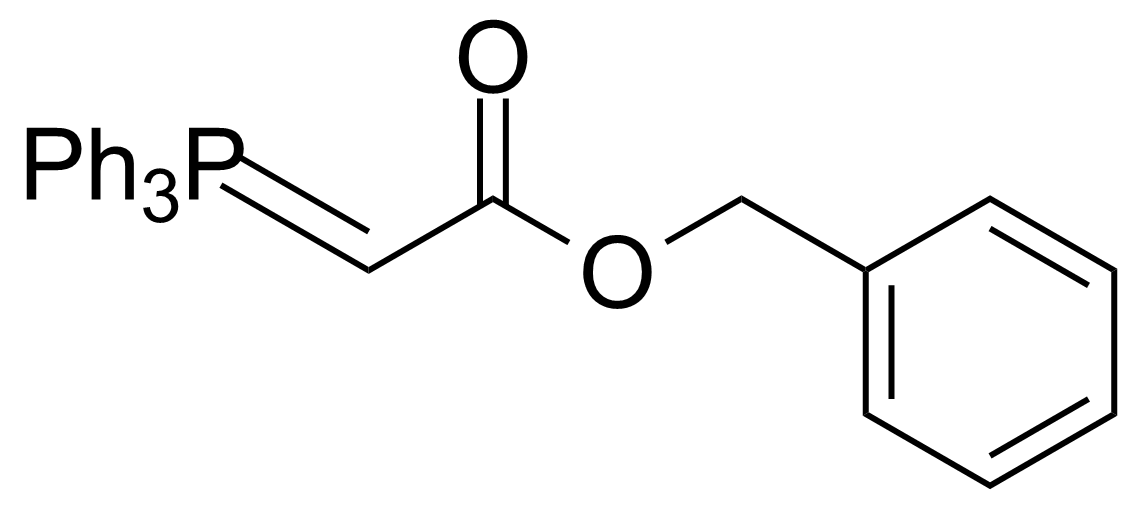 | [15097-38-8] | GEO-02511 | |
| New | 4-Bis(3,5-ditert-butylphenyl)phosphanylbutyl-bis(3,5-ditert-butylphenyl)phosphane | 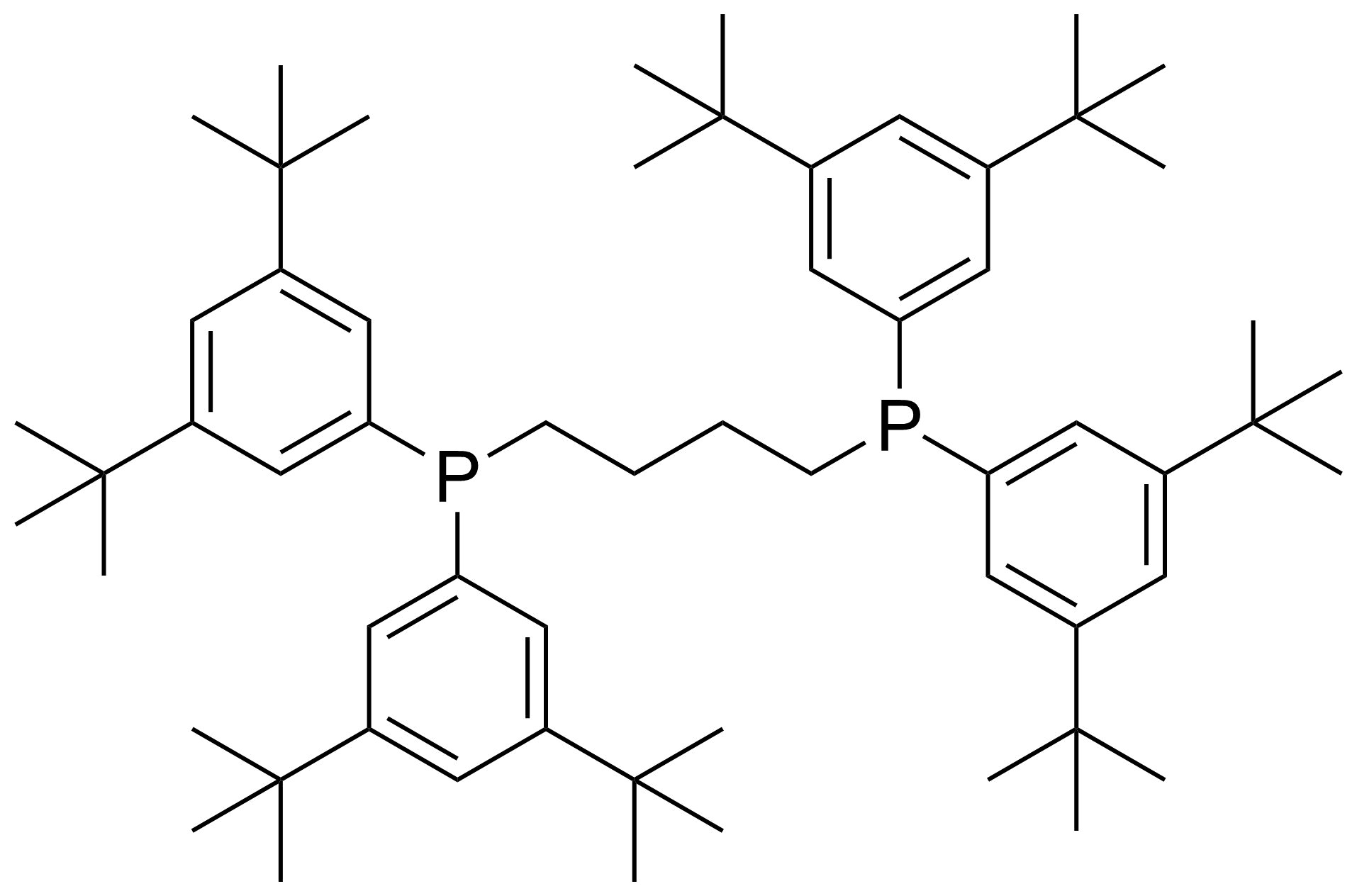 | [1800417-20-2] | GEO-04884 |
| Bis(dichlorophosphino)methane | 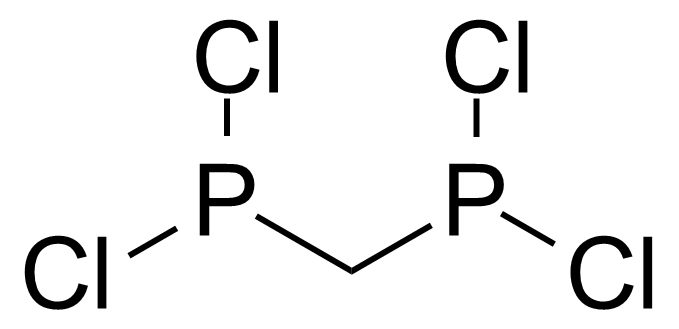 | [28240-68-8] | GEO-03420 | |
| Bis(diethylamino)chlorophosphine | 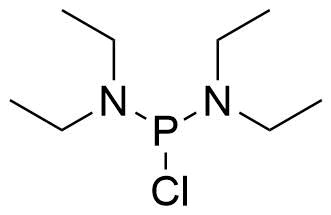 | [685-83-6] | GEO-04741 |