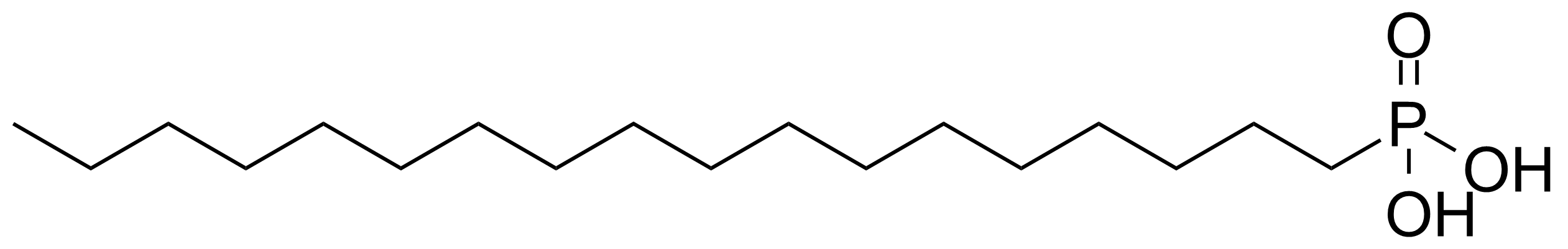
Octadecylphosphonic acid
CAS#[4724-47-4]
G-codeGEO-04391
EC number225-216-4
Molecular formulaC18H39O3P
Molecular weight334.48
Synonyms
Stearylphosphonic acid
For more information or to place an inquiry, please email us to
georganics@georganics.sk or use our contact form
Regulatory Information
This product has not been classified.
Product categorization
Main category
Second level
Description
Octadecylphosphonic acid is used in thermal paper for receipts, adding machines and tickets. The acid serves as a matrix….
Show full descriptionGeneral description and preparation:
Octadecylphosphonic acid (ODPA) [4724-47-4] is a white crystalline solid with the melting point of 93-95 °C.[1] It is a long chain phosphonic acid compound, irritating to eyes, respiratory system and skin. It is an amphiphilic molecule, possessing both hydrophilic and lipophilic properties. Octadecylphosphonic acid can be prepared by Arbuzov reaction via nucleophilic attack of a trialkyl phosphite to octadecyl bromide (stearyl bromide). Phosphonic acid is obtained after hydrolysis of appropriate phosphonate. Recently was reported the use of tris(trimethylsilyl) phosphite [P(OTMS)3]. Treatment of H3PO3 with 1 equiv of stearyl bromide and excess BSTFA followed by hydrolysis with methanol and water gives stearylphosphonic acid in nearly quantitative yield.[2]Application of Octadecylphosphonic acid:
ODPA is used in thermal paper for receipts, adding machines and tickets. The acid serves as a matrix. When heat liquefies the acid, a reaction between the dye and the acid occurs. This reaction causes the colour of the thermal paper to change.[3] It acts as surfactant, emulsifier, dispersant and chelating agent. It is utilized for proteomics research. In addition to this, it is used to prepare modified mesoporous magnetic nano particles through Lewis acid/base interaction.[4] ODPA is mostly known for its ability to form self-assembled monolayers (SAMs) on a large variety of substrates.[5] It has demonstrated potential for deterring bacterial attachment to titanium.[6]Product categorization (Chemical groups):
Main category: Second level: _______________________________________________________________________ [1] V. A. Kuimov, S. F. Malysheva, N. A. Belogorlova, A. I. Albanov, N. K. Gusarova, B. A. Trofimov Eur. J. Org. Chem. 2021, 2021 (10), 1596. doi:10.1002/ejoc.202100067 [2] N. Zhang, J. E. Casida J. Org. Chem. 2001, 66 (1), 327. doi:10.1021/jo0013608 [3] K. Tsutsui, T. Y. Sato Jpn. J. Appl. Phys. 1994, 33, 5925. doi:10.1143/JJAP.33.5925 [4] J. Ding, Q. Gao, D. Luo, Z. G. Shi, Y. Q. Feng J. Chromatogr. A 2010, 1217 (47), 7351. doi:10.1016/j.chroma.2010.09.074 [5] G. N. Fontes, A. Malachias, R. Magalhães-Paniago, B. R. A. Neves Langmuir 2003, 19 (8), 3345. doi:10.1021/la0267847 [6] L. Azizova, D. Morgan, J. Rowlands, E. Brousseau, T. Kulik, B. Palianytsia, J. P. Mansell, J. Birchal, T. Wilkinson, A. Sloah, W. N. Ayre Appl. Surf. Sci. 2022, 604, 154462. doi:10.1016/j.apsusc.2022.154462
Similar products
| Product name | Structure | CAS# | G-code | |
|---|---|---|---|---|
| 2-Aminoethyl dihydrogen phosphate | 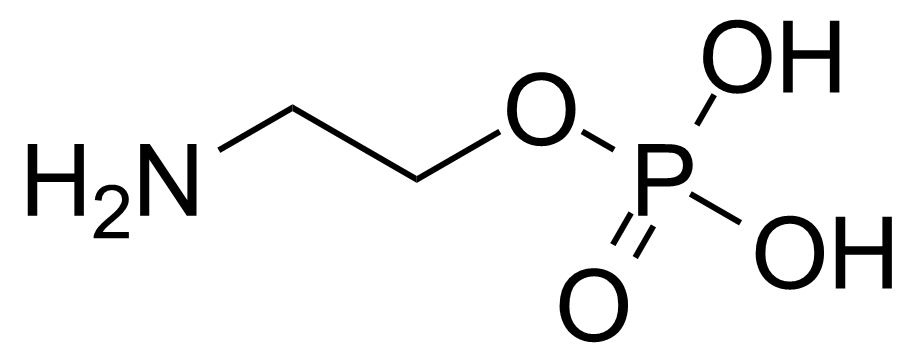 | [1071-23-4] | GEO-00129 | |
| 2-Aminoethyl dihydrogen phosphate magnesium salt (2:1) | 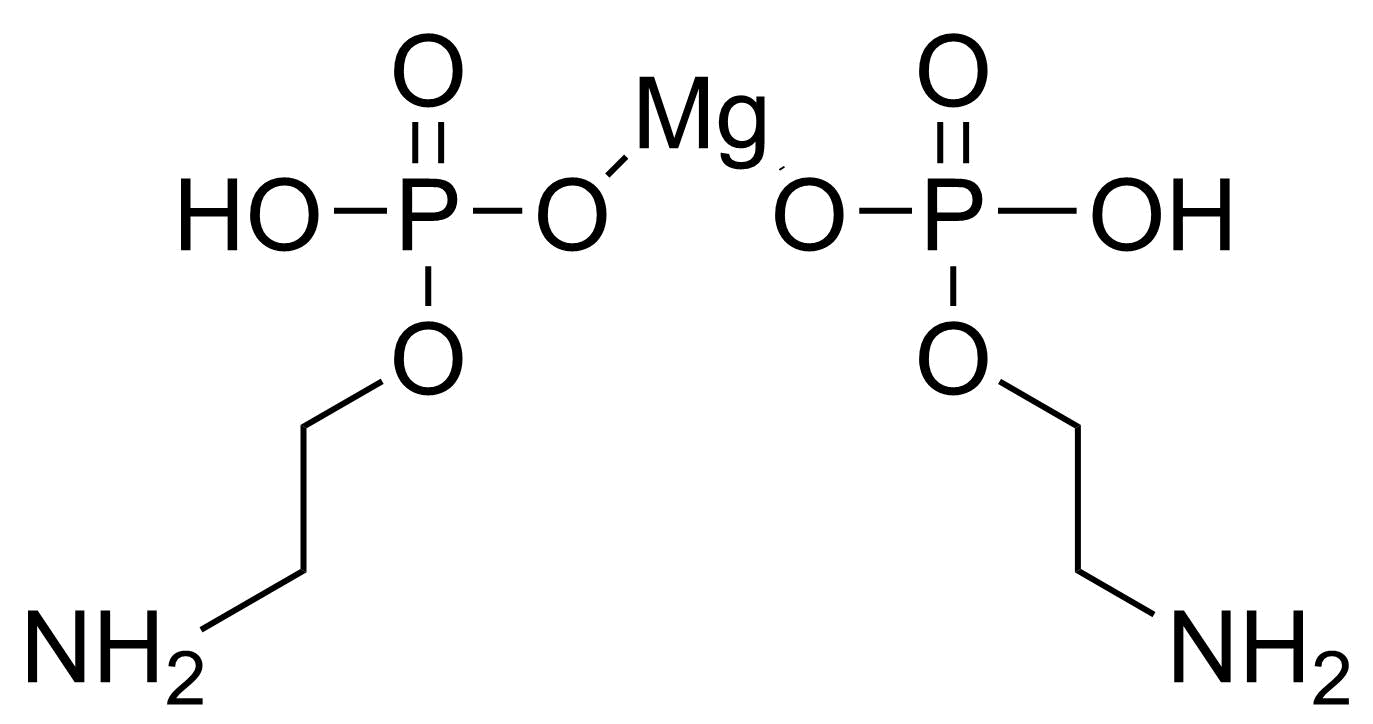 | N/A | GEO-04286 | |
| (Aminomethyl)phosphonic acid | 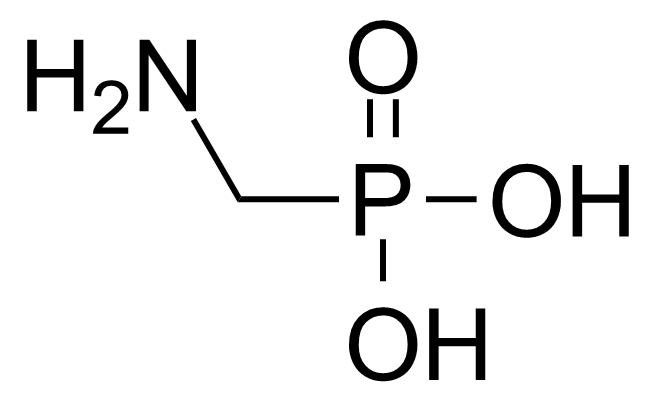 | [1066-51-9] | GEO-00169 | |
| (3-Aminopropyl)phosphonic acid | 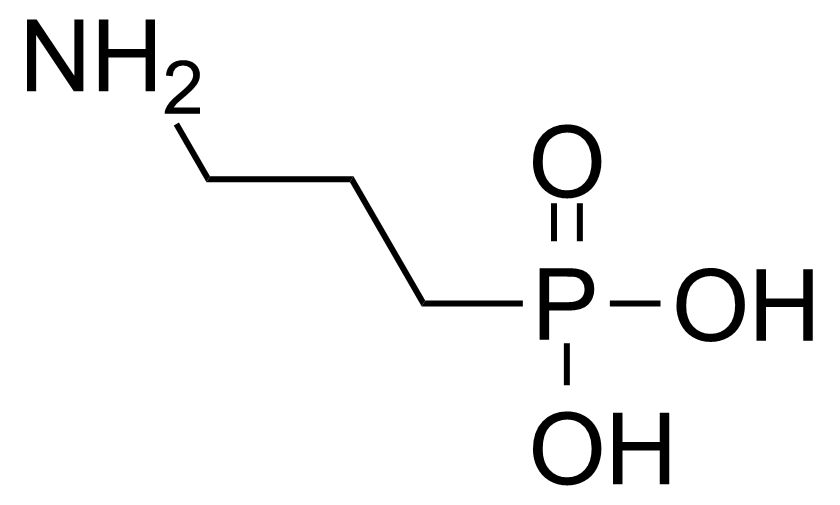 | [13138-33-5] | GEO-00218 | |
| Benzo[d]isoxazol-3-yl diphenyl phosphate | ![Structure of Benzo[d]isoxazol-3-yl diphenyl phosphate](https://georganics.sk/wp-content/uploads/2021/05/GEO-00266_Benzodisoxazol-3-yl_diphenyl_phosphate.png) | [94820-31-2] | GEO-00266 | |
| Benzyl dimethylphosphonoacetate | 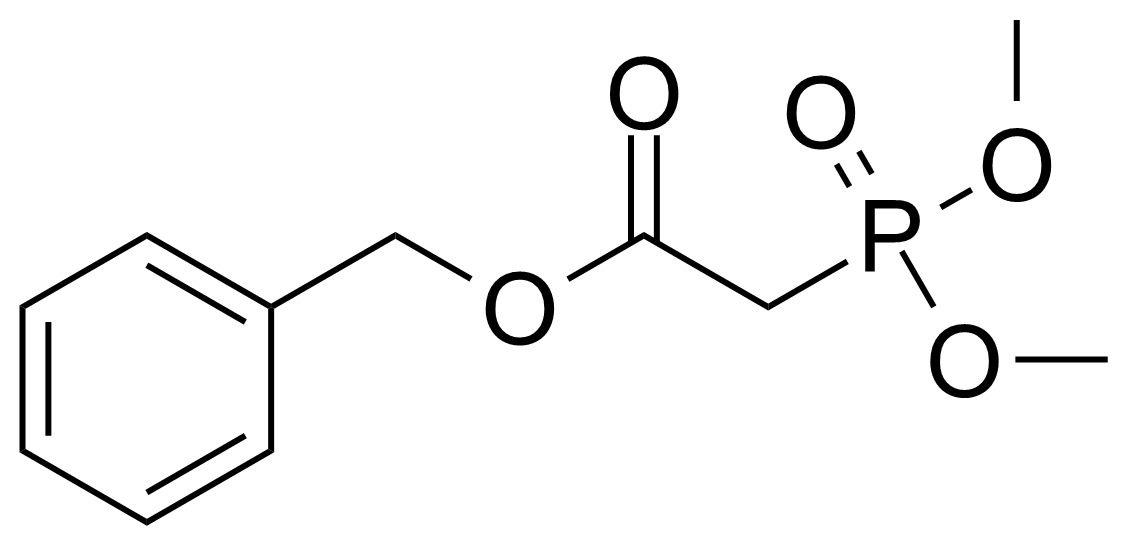 | [57443-18-2] | GEO-02882 | |
| Benzyl(triphenylphosphoranylidene)acetate | 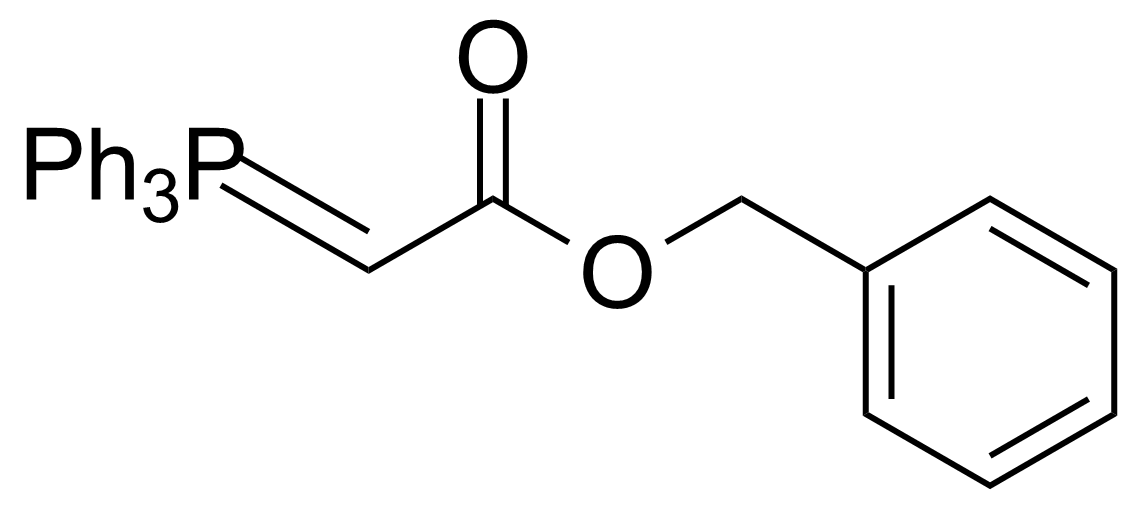 | [15097-38-8] | GEO-02511 | |
| New | 4-Bis(3,5-ditert-butylphenyl)phosphanylbutyl-bis(3,5-ditert-butylphenyl)phosphane | 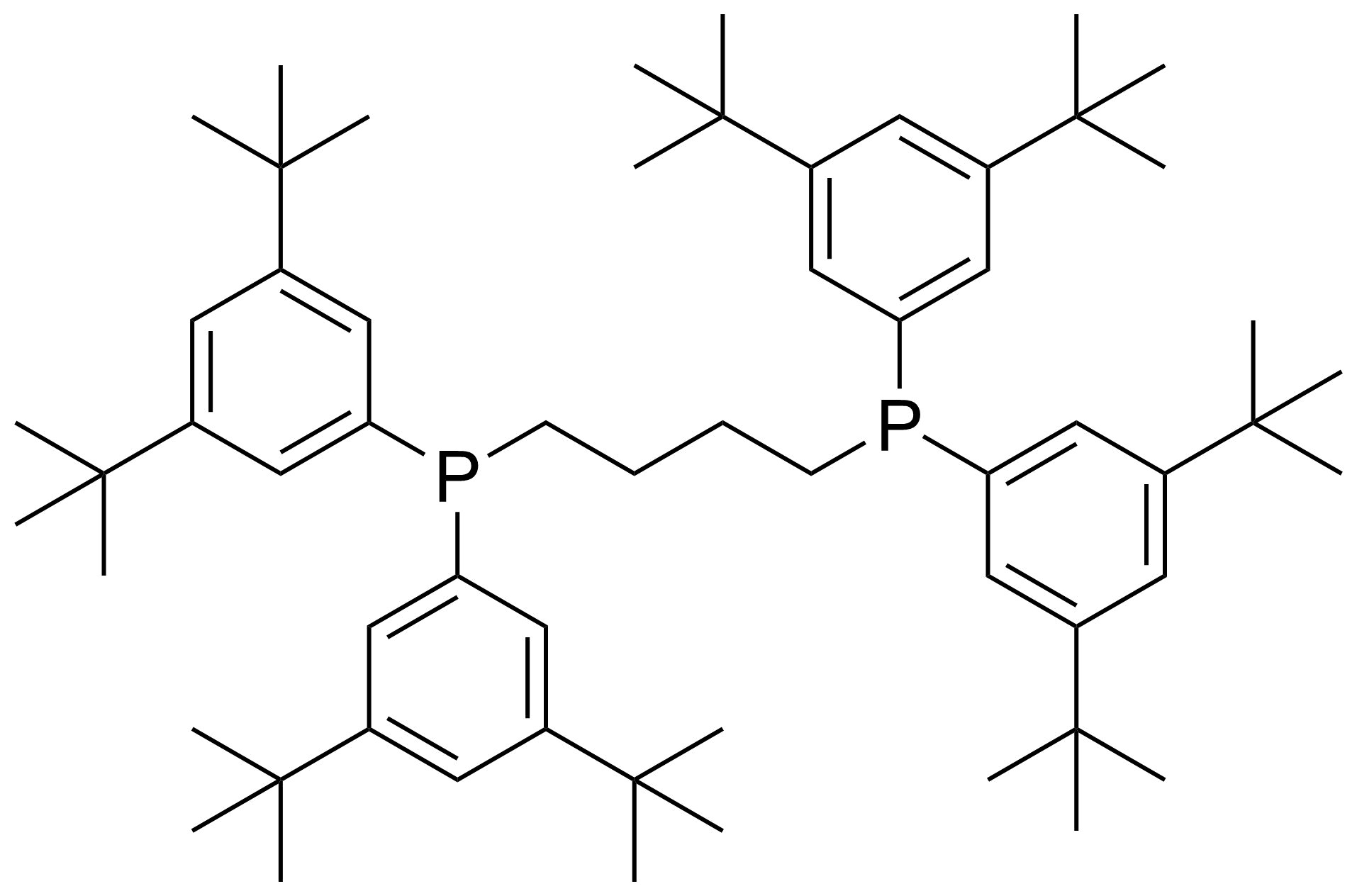 | [1800417-20-2] | GEO-04884 |
| Bis(dichlorophosphino)methane | 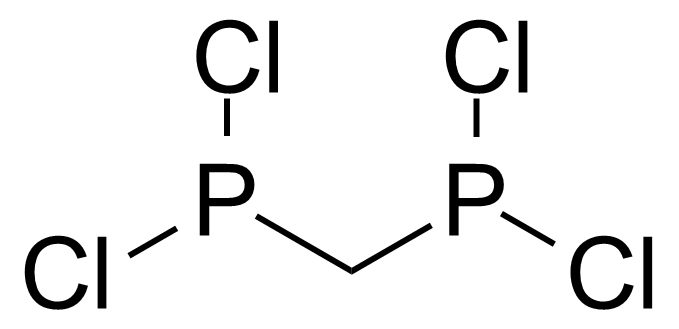 | [28240-68-8] | GEO-03420 | |
| Bis(diethylamino)chlorophosphine | 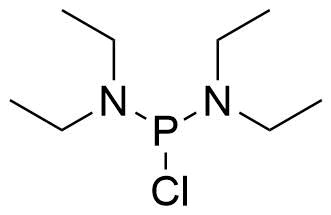 | [685-83-6] | GEO-04741 |