 May 18, 2023
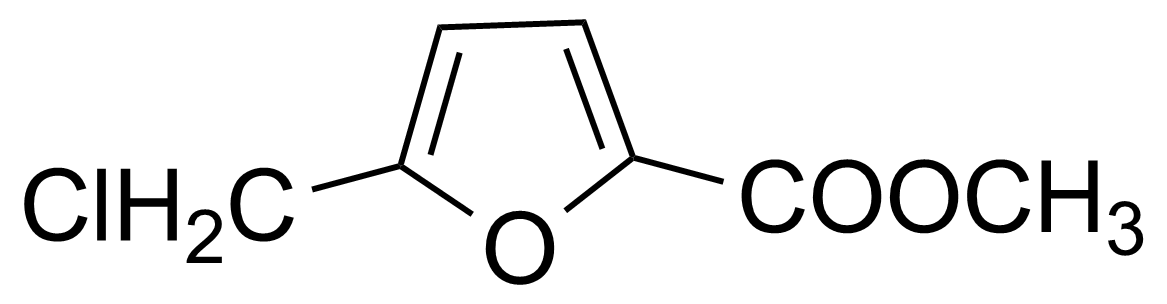
May 18, 2023Methyl 5-(chloromethyl)-2-furoate – preparation and application
Methyl 5-(chloromethyl)-2-furoate [2144-37-8] is a white to yellow crystalline solid with the melting point of 29-30 °C. [1] At higher temperatures it is a clear oil with the boiling point of 114-116 °C/3 mbar. It is insoluble in water and soluble in the usual organic solvents. It darkens it color after long standing.[2]
It can be prepared by Blanc type chloromethylation of commercially available methyl furan-2-carboxylate with paraformaldehyde, zinc chloride and anhydrous hydrogen chloride.[3] Another possibility of preparation is chlorination of hydroxymethyl derivative, which can be obtained from D-glucono-δ-lactone.[4]
Application of Methyl 5-(chloromethyl)-2-furoate:
Methyl 5-(chloromethyl)furan-2-carboxylate is used as a versatile building block in the synthesis of various biologically relevant compounds. It was used in the synthesis conjugated hydroxamic acids as a potent anti-tumor histone deacetylase (HDAC) inhibitors.[5] It also served as a starting material in the synthesis of furan-2-carboxamide analogues as novel positive allosteric modulators of N-Methyl-D-aspartate receptors.[6] It was used in the synthesis of novel guanidine-derived non-peptide gonadotropin releasing hormone receptor antagonists that may serve as useful therapeutic agents for treating hormone-dependent pathologies including hormone-dependent prostate and breast cancer.[7]
Product categorization (Chemical groups):
Main category:
Second level:
Third level:
_______________________________________________________________________
[2] A. L. Mndzhoian Methyl 5-(Chloromethyl)-2-Furoate. In: Syntheses of Heterocyclic Compounds 1959, 31, Springer doi:10.1007/978-1-4757-6658-5_13
[3] S. Nielek, T. Liesak J. Prakt. Chem. 1988, 330, 825. doi:10.1002/prac.19883300523
[4] M. J. Pedersen, C. M. Pedersen Synthesis of precursors of 2,5-furandicarboxylic acid 2019, Københavns Universitet, WO2019170204A1
[5] H. Su, A. Nebbioso, V. Carafa, Y. Chen, B. Yang, L. Altucci, Q. You Bioorg. Mech. Chem. 2008, 16 (17), 7992. doi:10.1016/j.bmc.2008.07.066
[6] Z. Li, G. Cai, F. Fang, W. Li, M. Fan, J. Lian, Y. Qiu, X. Xu, X. Lv, Y. Li, R. Zheng, Y. Wang, Z. Li, G. Zhang, Z. Liu, Z. Huang, L. Zhang J. Med. Chem. 2021, 64 (9), 5551. doi:0.1021/acs.jmedchem.0c02018
[7] D. R. Luthin, Y. Hong, V. P. Pathak, G. Paderes, K. D. Nared-Hood, M. A. Castro, H. Vazir, H. Li, E. Tompkins, L. Christie, J. M. May, M. B. Anderson Bioorg. Med. Chem. Lett. 2002, 12 (23), 3467. doi:10.1016/S0960-894X(02)00755-2