 February 01, 2022
February 01, 2022Diethyl phosphate – general description and application
General description of Diethyl phosphate:
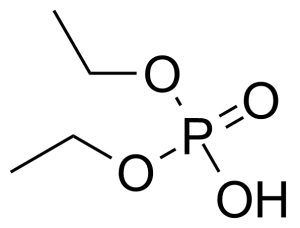 Diethyl phosphate (DEP) [598-02-7], diethyl hydrogen phosphate or diethyl phosphoric acid is a colorless oil with the boiling point of 80 °C at 0.7 mm Hg.[1] Despite its low toxitcity, it is a combustible liquid that can cause severe skin burns and eye damage. It is soluble in common organic solvents and water.
Diethyl phosphate (DEP) [598-02-7], diethyl hydrogen phosphate or diethyl phosphoric acid is a colorless oil with the boiling point of 80 °C at 0.7 mm Hg.[1] Despite its low toxitcity, it is a combustible liquid that can cause severe skin burns and eye damage. It is soluble in common organic solvents and water.
DEP can be prepared from diethyl chlorophosphate using benzaldoxime via diethyl benzaldimino phosphate intermediate which in turn decomposes to diethyl phosphate and benzonitrile. Tert-butanol can be also used instead of benzaldoxime providing product however in a lower yield.[2] Another preparation method starts with the phosphorous trichloride using the ethanol followed by a treatment with pyridine and CCl4. Formed trichloroethyl ester is then converted to mixed anhydride with acetic acid and Et3N followed by acid hydrolysis.1
Application of Diethyl phosphate:
DEP is a product of metabolism and environmental degradation of a commonly used insecticide Chlorpyrifos and it is routinely measured in urine as biomarker of exposure.[3] Chronic exposure to DEP can cause thyroid-related hormone disorders.[4]
Product categorization (Chemical groups):
Main category:
Regulatory Information:
- Transport Information: CORROSIVE LIQUID, ACIDIC, ORGANIC, N.O.S. UN3265 8/ PG III
- Signal Word: Danger
- Hazard Statements:
- H302: Harmful if swallowed
- H312: Harmful in contact with skin
- H314: Causes severe skin burns and eye damage
- H318: Causes serious eye damage
- Precautionary Statements:
- P261: Avoid breathing dust/fume/gas/mist/vapours/spray:
- P280: Wear protective gloves/protective clothing/eye protection/face protection:
- P301+330+331: IF SWALLOWED: Rinse mouth. Do NOT induce vomiting
- P302+352: IF ON SKIN: Wash with soap and water
- P305+351+338: IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses if present and easy to do – continue rinsing
- P310: Immediately call a POISON CENTER or doctor/physician:
_______________________________________________________________________
[2]T. Mukaiyama, T. Fujisawa Bull. Chem. Soc. Jpn. 1961, 34, 812.
[3]Ch. Timchalk, A. Busby, J. A. Campbell, L. L. Deedham, D. B. Barr Toxicology 2007, 237, 145.
[4]F. W. Yang, G. P. Zhao, F. Z. Ren, G. F. Pang, Y. X. Li Environ. Int. 2020, 135, 105383.