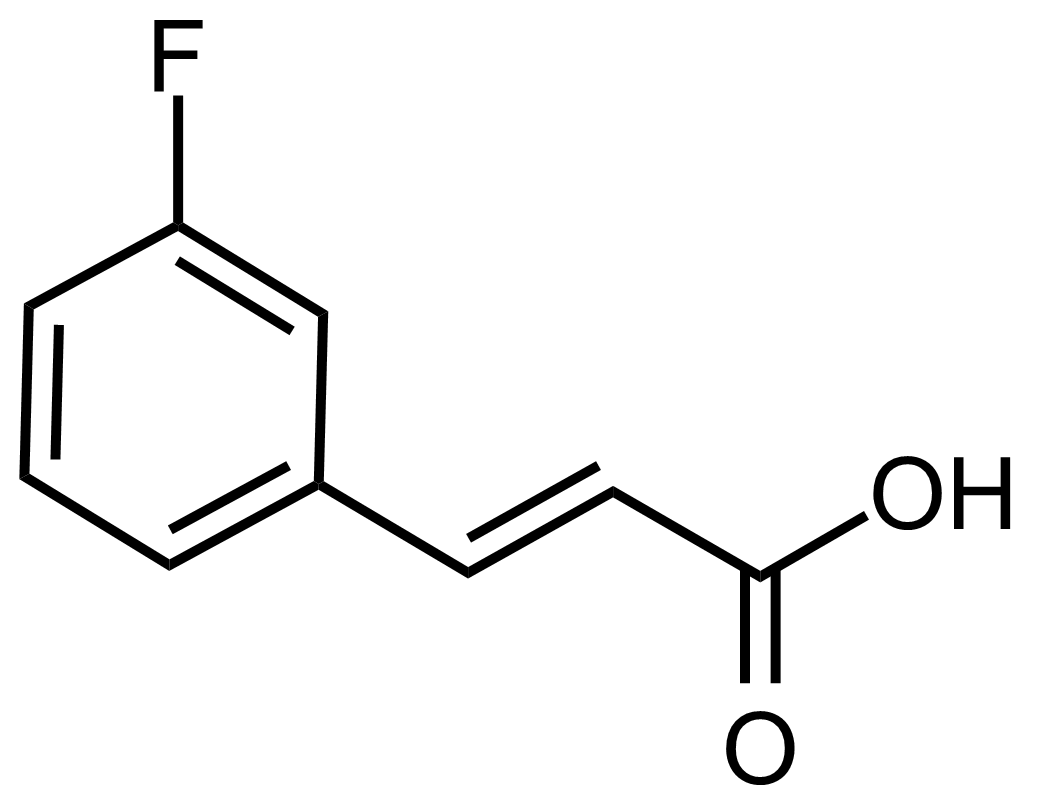
(E)-3-(3-Fluorophenyl)acrylic acid
3-(3-fluorophenyl)prop-2-enoic acid ; (E)-m-Fluorocinnamic acid ; 3-Fluorocinnamic acid ; ; trans-3-Fluorocinnamic acid ; trans-3-(3-Fluorophenyl)propenoic acid ; m-Fluorocinnamic acid
For more information or to place an inquiry, please email us to
georganics@georganics.sk or use our contact form
Regulatory Information
Not regulated as a dangerous good.

H301 – Toxic if swallowed
H315 – Causes skin irritation
H319 – Causes serious eye irritation
H335 – May cause respiratory irritation
P261 – Avoid breathing dust/fume/gas/mist/vapours/spray:
P301+310 – IF SWALLOWED: Immediately call a POISON CENTER or doctor/physician
P305+351+338 – IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses if present and easy to do – continue rinsing
Product categorization
Description
(E)-3-(3-Fluorophenyl)acrylic acid [20595-30-6], 3-fluorocinnamic acid, or (2E)-3-(3-Fluorophenyl)-2-propenoic acid, is an organic acid belonging to acrylic acids group. In its pure form, it is a white crystalline solid with a melting point of 166,2-166,8 °C.[1] It is soluble in water and polar organic solvents…
Show full descriptionGeneral description of (E)-3-(3-Fluorophenyl)acrylic acid:
(E)-3-(3-Fluorophenyl)acrylic acid [20595-30-6], 3-fluorocin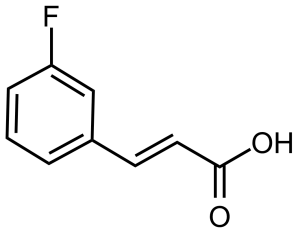 namic acid, or (2E)-3-(3-Fluorophenyl)-2-propenoic acid, is an organic acid belonging to acrylic acids group. In its pure form, it is a white crystalline solid with a melting point of 166,2-166,8 °C.[1] It is soluble in water and polar organic solvents. It is known that 3-fluorocinnamic acid can cause serious damage if swallowed and can be irritating in contact with skin or eyes (H301, H315, H319).[2]
namic acid, or (2E)-3-(3-Fluorophenyl)-2-propenoic acid, is an organic acid belonging to acrylic acids group. In its pure form, it is a white crystalline solid with a melting point of 166,2-166,8 °C.[1] It is soluble in water and polar organic solvents. It is known that 3-fluorocinnamic acid can cause serious damage if swallowed and can be irritating in contact with skin or eyes (H301, H315, H319).[2]Preparation:
3-Fluorocinnamic acid can be prepared by Knowenagel-Doebner reaction of malonic acid or malonic ester with corresponding aldehyde. [3] It is also formed as side product in kinetic resolution of β-phenylalanine derivatives via selective conversion of a single enantiomer to the corresponding acrylic acid. [4] Recent research also showed that it can be prepared by Suzuki coupling of corresponding boronic acid with carbon dioxide. [5] _______________________________________________________________________Similar products
| Product name | Structure | CAS# | G-code | |
|---|---|---|---|---|
| 2-Acetamido-3,4,6-tri-O-acetyl-2-deoxy-alpha-D-glucopyranosyl chloride | 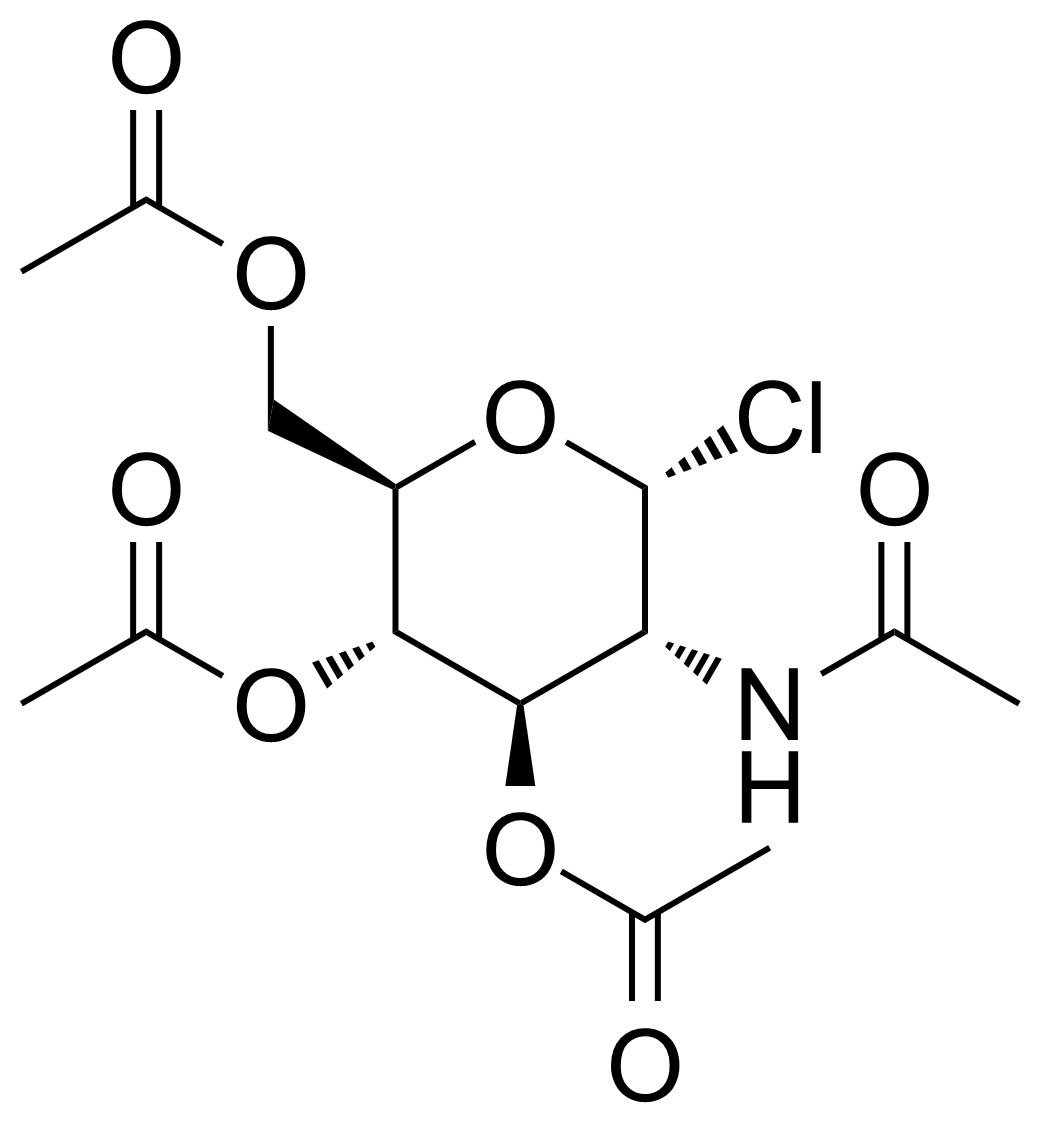 | [3068-34-6] | GEO-03374 | |
| Acetochloro-beta-D-glucose | 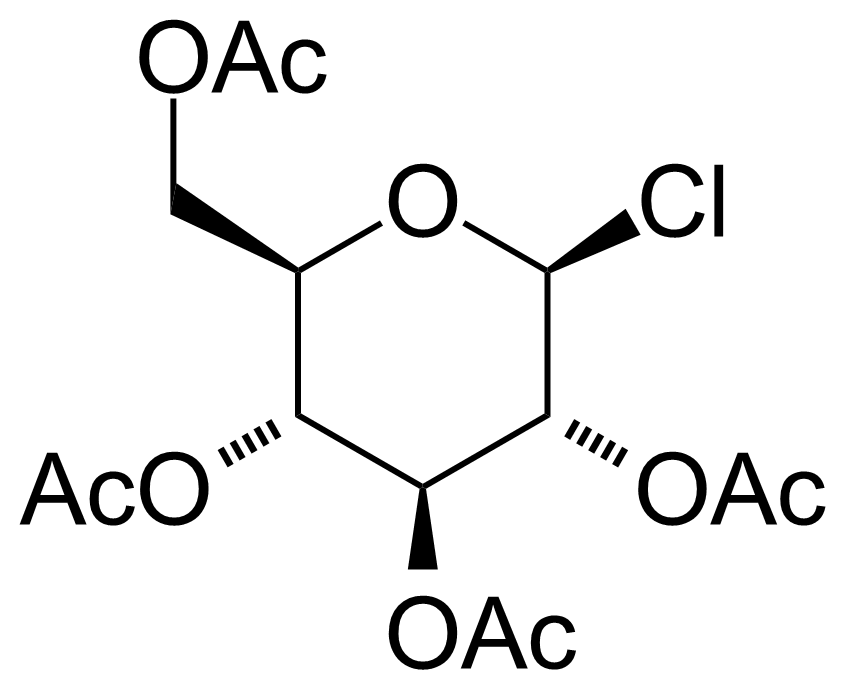 | [4451-36-9] | GEO-00008 | |
| 2-Acetyl-4-bromothiophene | 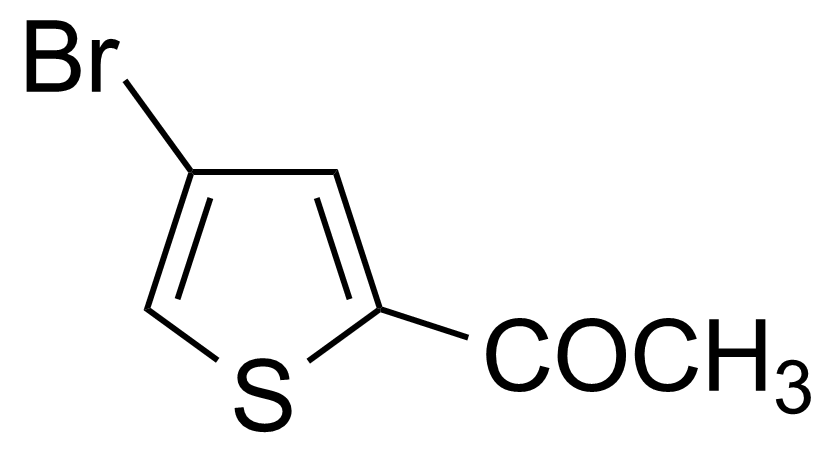 | [7209-11-2] | GEO-00023 | |
| 5-Amino-2-bromobenzoic acid | 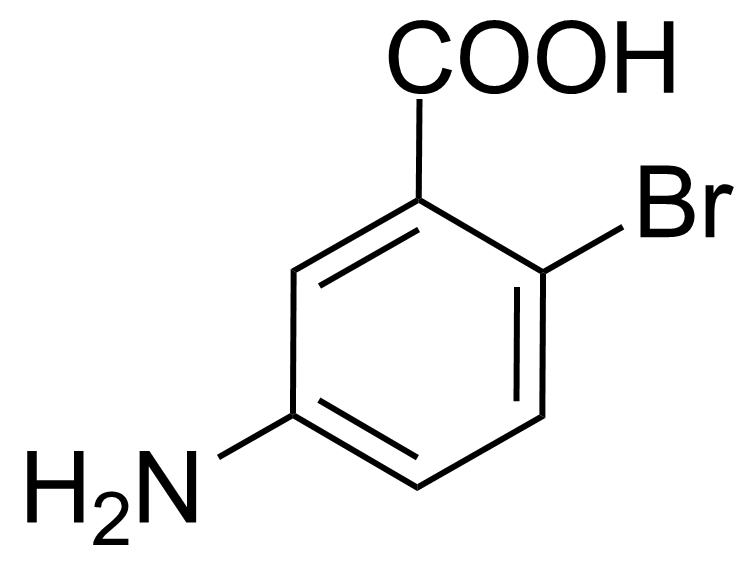 | [2840-02-0] | GEO-00082 | |
| 2-Amino-6-bromobenzothiazole | 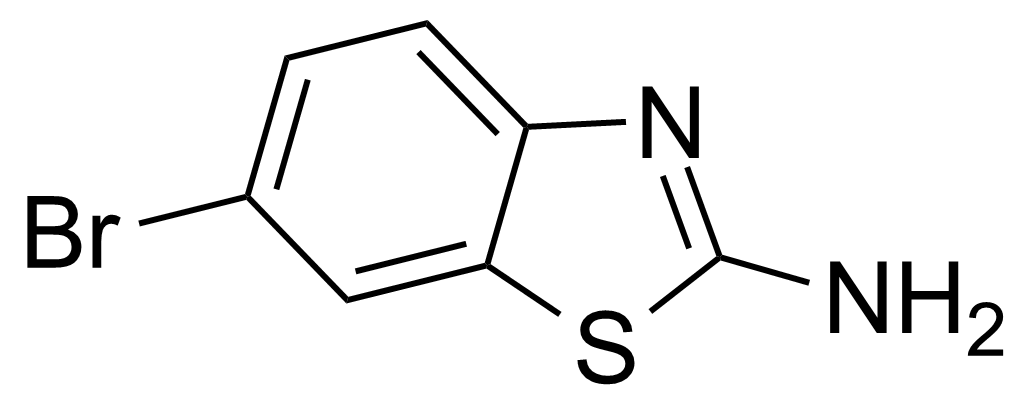 | [15864-32-1] | GEO-00083 | |
| 6-Amino-5-bromo-1-methyluracil monohydrate | 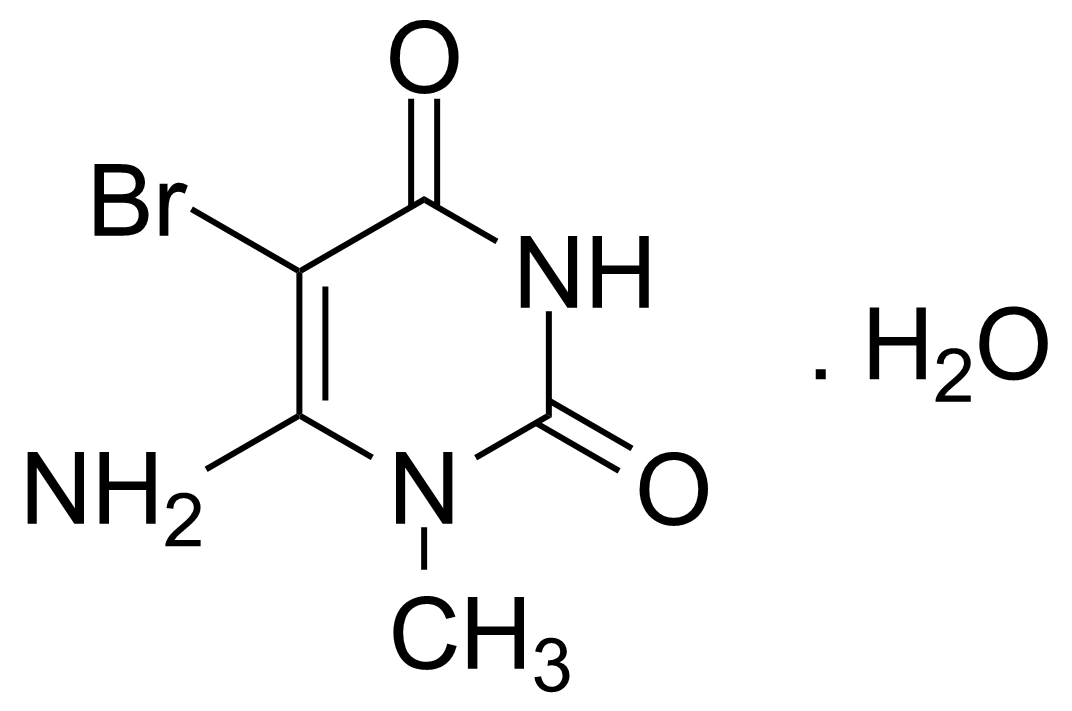 | [14094-37-2] | GEO-00087 | |
| 2-Amino-5-chlorobenzonitrile | 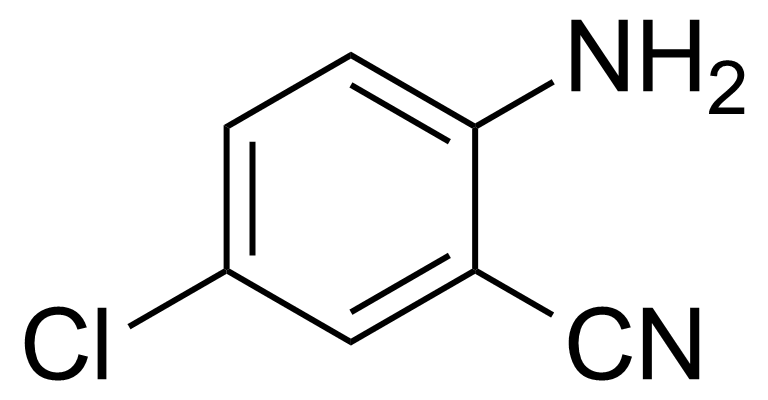 | [5922-60-1] | GEO-00097 | |
| 2-Amino-4-chlorobenzothiazole | 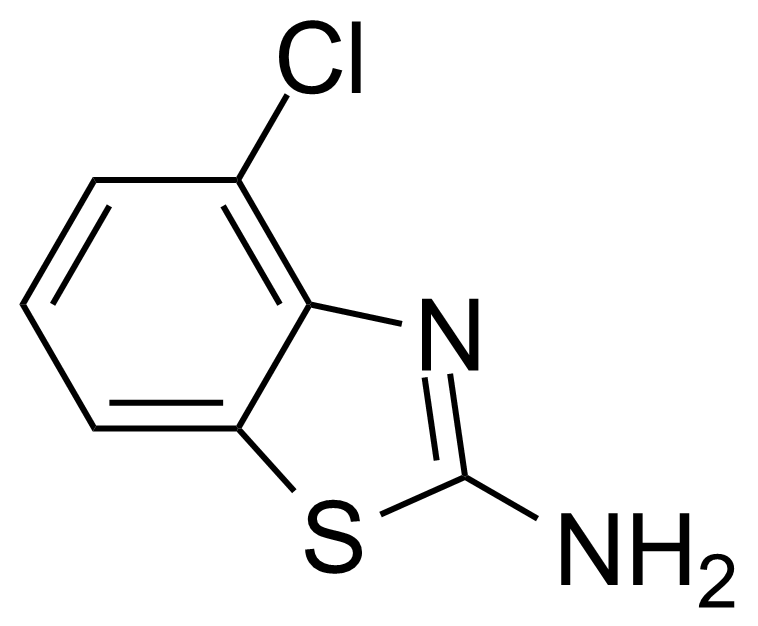 | [19952-47-7] | GEO-00099 | |
| 2-Amino-6-chlorobenzothiazole | 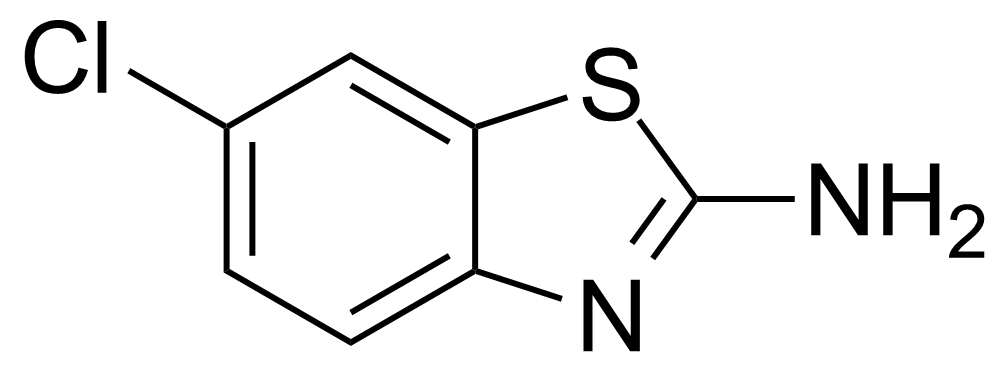 | [95-24-9] | GEO-02880 | |
| 2-Amino-4-chloro-3-iodopyridine | 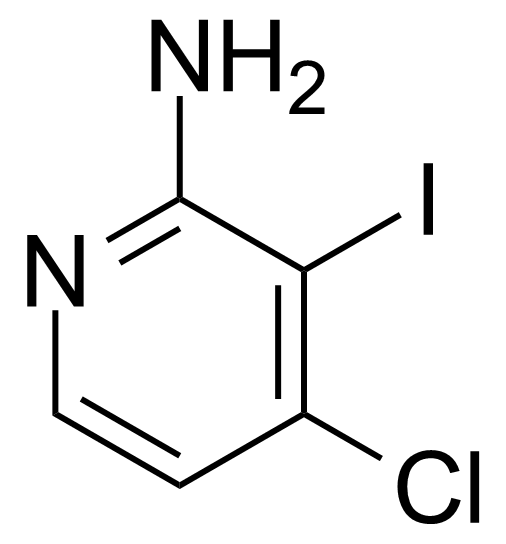 | [417721-69-8] | GEO-03750 |