L-Pyroglutamic acid
L-Pyroglutamic acid ; 5-Oxoproline ; 5-Oxopyrrolidine-2-carboxylic acid ; L-5-Pyrrolidone-2-carboxylic acid ; (S)-(-)-2-Pyrrolidone-5-carboxylic acid ; (2S)-5-hydroxy-3,4-dihydro-2H-pyrrole-2-carboxylic acid ; (2S)-5-Oxopyrrolidine-2-carboxylic acid ; (S)-5-Oxo-2-pyrrolidine carboxylic acid ; (S)-5-oxopyrrolidine-2-carboxylic acid ; 2-Aminopentanedioic acid ; 5-Oxo-L-proline ; L-2-Pyrrolidone-5-carboxylic acid ; Pidolic acid ; L-Pyrrolidonecarboxylic acid
For more information or to place an inquiry, please email us to
georganics@georganics.sk or use our contact form
Regulatory Information
Not a hazardous substance or mixture according to Regulation (EC) No. 1272/2008.
Product categorization
Description
L-Pyroglutamic acid is a useful chemical compound with a variety of research applications. We are pleased to offer high quality L-Pyroglutamic acid in various sizes (for research, pilot-scale, or production applications) from milligrams to multi-kilogram batches, making it easy for you to choose the right amount to suit your needs.
Show full descriptionPreparation of L-pyroglutamic acid:
PCA can be easily obtained by direct thermal dehydration of L-glutamic acid in water under reflux.[3]Application of L-pyroglutamic acid:
In neurology, the brain-boosting effects of PCA were discovered in 1984. It plays an important role in the preservation and activity of the key neurotransmitters acetylcholine, gamma-aminobutyric acid (GABA) and glutamic acid.[4] It readily passes the blood-brain barrier to stimulate cognitive-enhancing function in rats[5] and reduce age-associated memory decline in humans.[6] In drug delivery, PCA esters can be used as dermal penetration enhancers for therapeutic agents having poor skin permeation or hair growth agents.[7] In chemical synthesis, it has been used as a versatile chiral building block in asymmetric synthesis of alkaloids, pharmaceuticals, and many other natural products.[8],[9] PCA is also known as a novel organic nonlinear optical (NLO) material for tunable UV harmonic generation down to 266 nm at room temperature.[10]Product categorization (Chemical groups):
Main category: Second level: Third level: _______________________________________________________________________Similar products
| Product name | Structure | CAS# | G-code | |
|---|---|---|---|---|
| 3-Acetoxybenzo[b]furan | ![Structure of 3-Acetoxybenzo[b]furan](https://georganics.sk/wp-content/uploads/2021/05/GEO-00011_3-Acetoxybenzobfuran.png) | [93680-80-9] | GEO-00011 | |
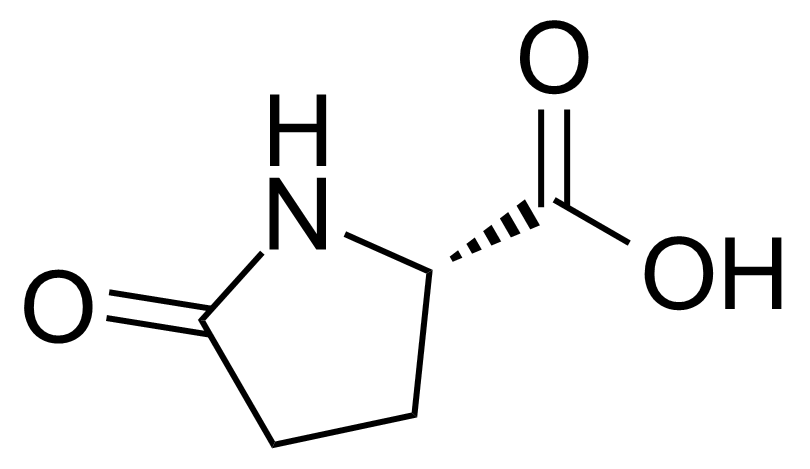
| 1-Adamantaneacetic acid | 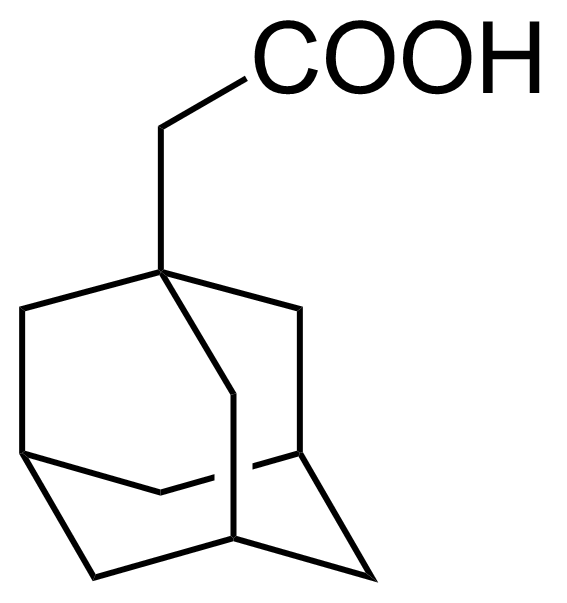 | [4942-47-6] | GEO-00049 | |
| 5-Amino-2-bromobenzoic acid | 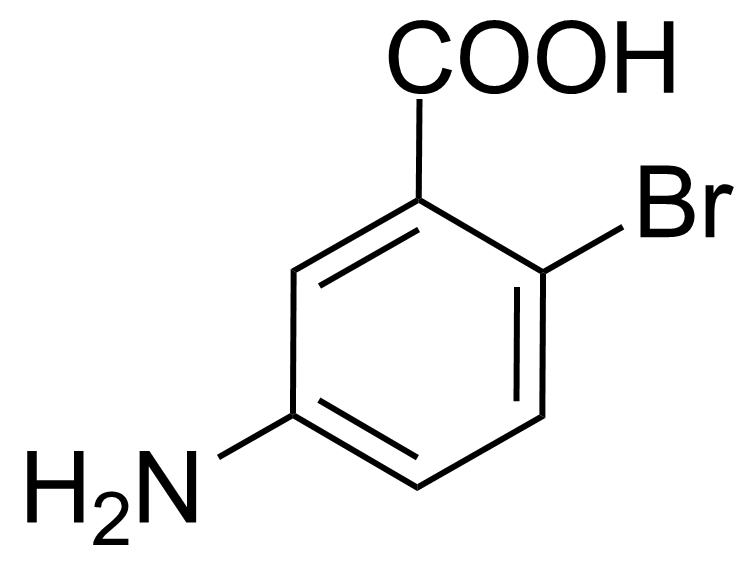 | [2840-02-0] | GEO-00082 | |
| (2R,4S)-2-Amino-4-cyclohexyl-4-hydroxybutanoic acid |  | [] | GEO-02717 | |
| 4-Amino-3-hydroxybenzoic acid | 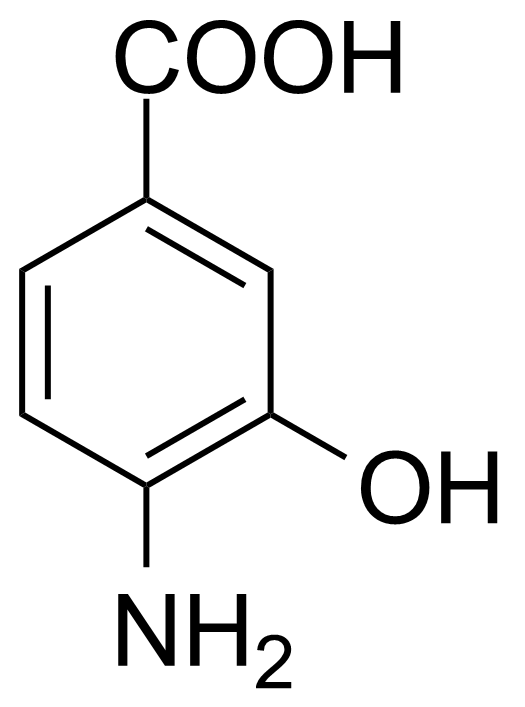 | [2374-03-0] | GEO-00141 | |
| (2R,4S)-2-Amino-4-hydroxy-4-phenylbutanoic acid | 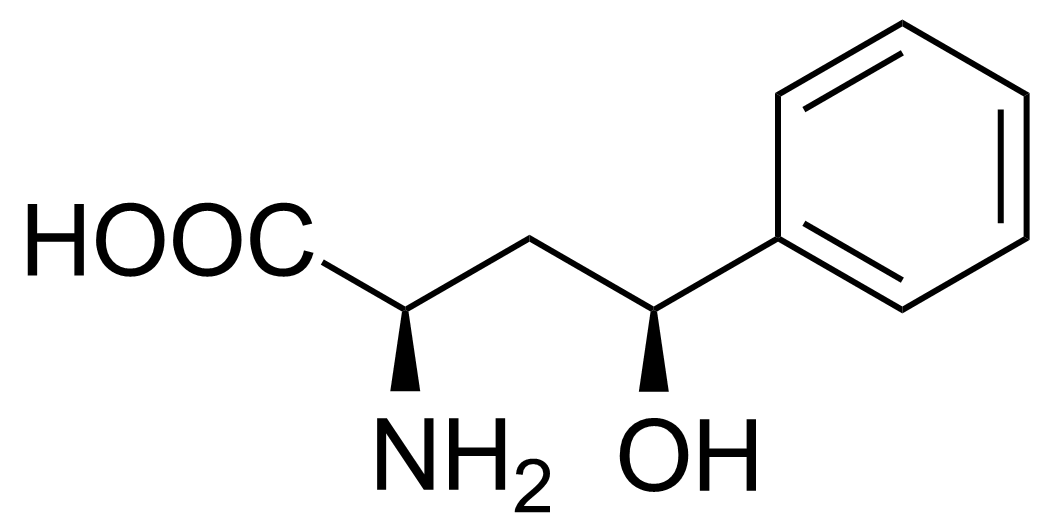 | [] | GEO-02718 | |
| (R)-2-Amino-2-((1S,2R)-1-hydroxy-1,2,3,4-tetrahydronaphthalen-2-yl)acetic acid | 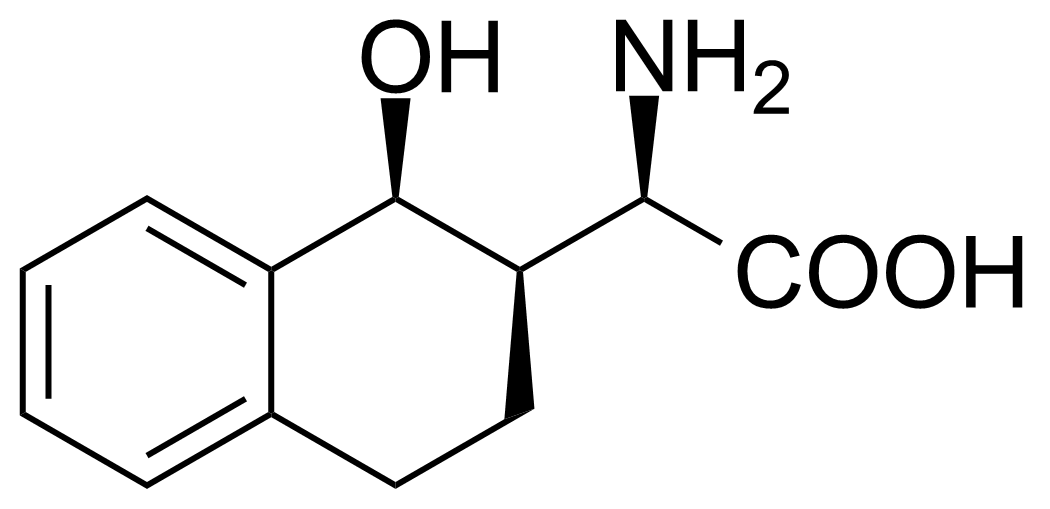 | [] | GEO-02719 | |
| 4-Amino-3-methoxybenzoic acid | 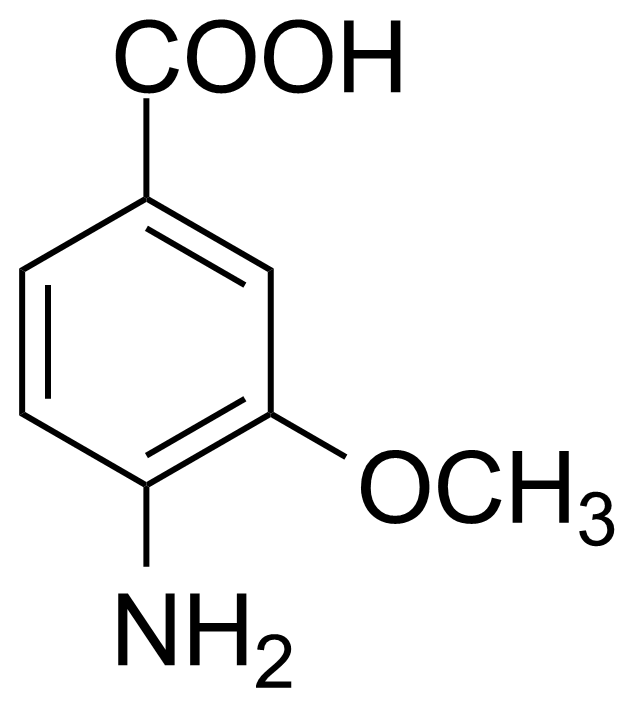 | [2486-69-3] | GEO-00154 | |
| 2-Amino-5-methylbenzoic acid | 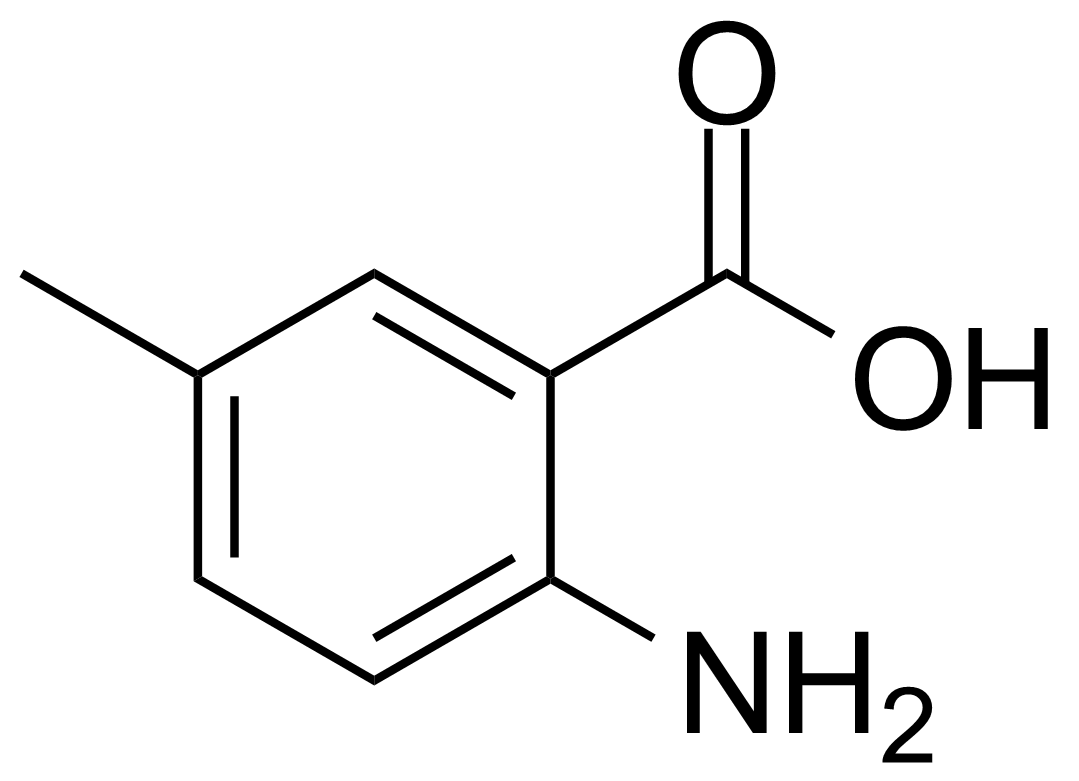 | [2941-78-8] | GEO-02910 | |
| 6-Amino-2-naphthoic acid | 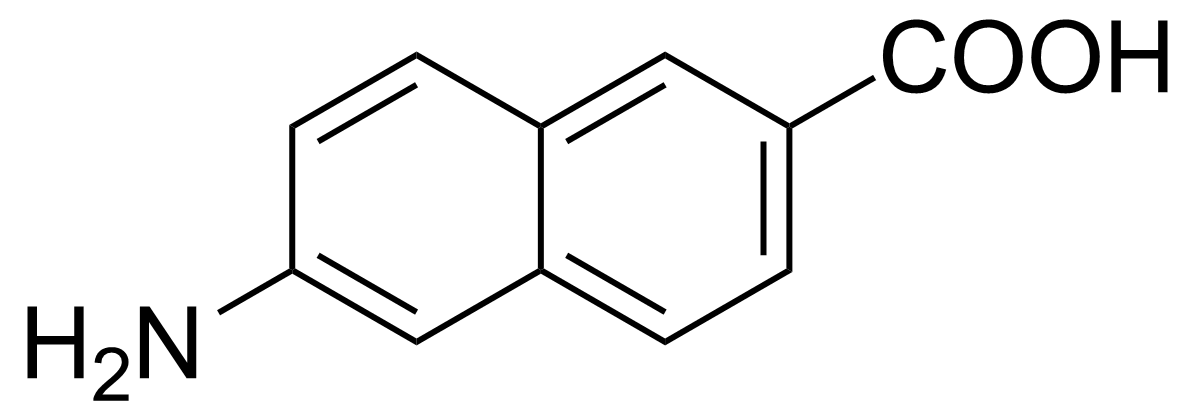 | [116668-47-4] | GEO-00179 |