Isocyanates
Isocyanates are a class of compounds that contain isocyanate group with the formula R−N=C=O. They are usually produced from amines by phosgenation via intermediacy of a carbamoyl chloride. Another route to isocyanates entails addition of isocyanic acid to alkenes or displacement reactions involving alkyl halides and alkali metal cyanates. Three rearrangement reactions involving nitrenes give isocyanates, Schmidt reaction, Curtius and Lossen rearrangement. As a good electrophiles they are reactive toward a variety of nucleophiles including alcohols, amines, and even water. They can undergo cyclization reactions and Hofmann rearrangement. Diisocyanates are widely used in industrial production of polyurethanes, organic polymers with application as different type of foams, surface coatings, adhesives, and fibres.
| Product name | Structure | CAS# | G-code | |
|---|---|---|---|---|
| Trichloroacetyl isocyanate | 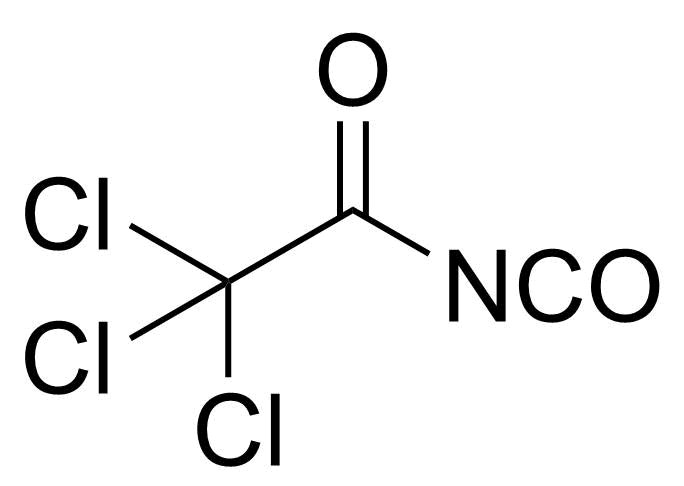 | [3019-71-4] | GEO-02352 | |
| 4-(Trifluoromethyl)benzyl isocyanate | 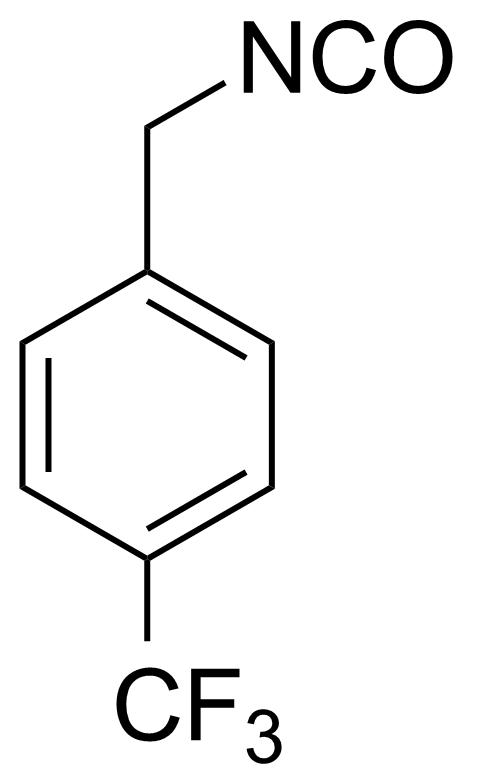 | [102422-55-9] | GEO-02377 | |
| 4-(Trifluoromethyl)phenyl isocyanate | 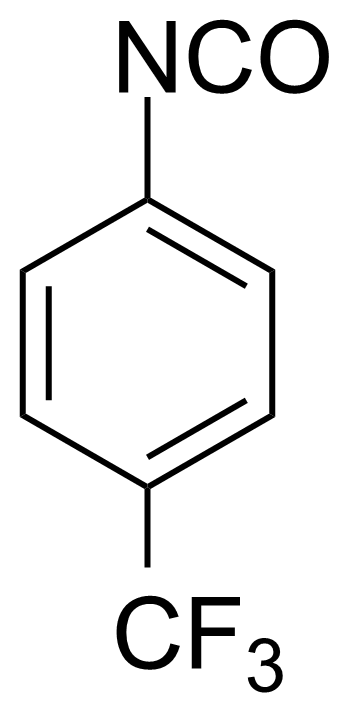 | [1548-13-6] | GEO-02386 | |
| 2-(Trifluoromethyl)phenyl isocyanate | 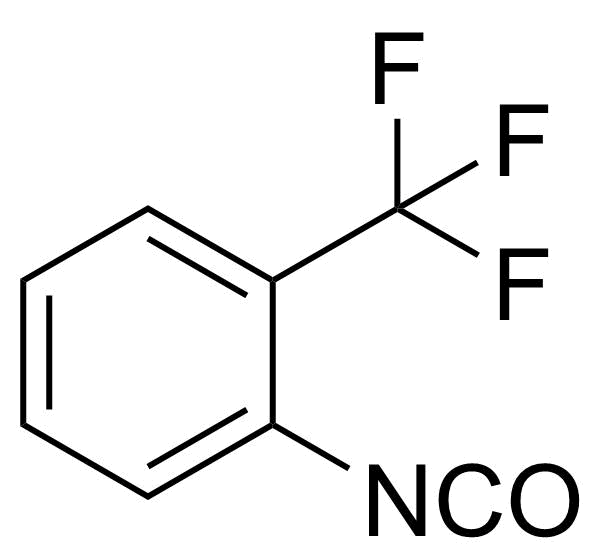 | [2285-12-3] | GEO-02396 | |
| 3-(Trifluoromethyl)phenyl isocyanate | 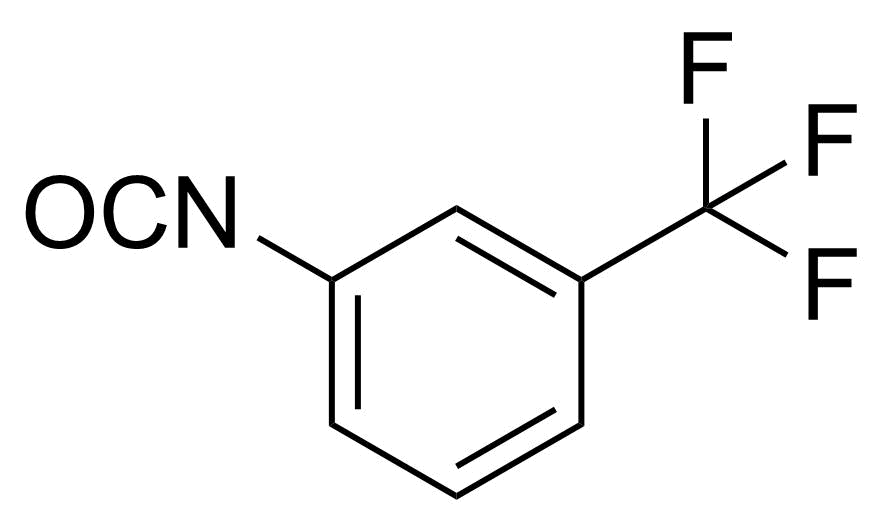 | [329-01-1] | GEO-02397 | |
| 2,4,6-Trifluorophenyl isocyanate |  | [50528-80-8] | GEO-02402 | |
| 3,4,5-Trimethoxyphenyl isocyanate | 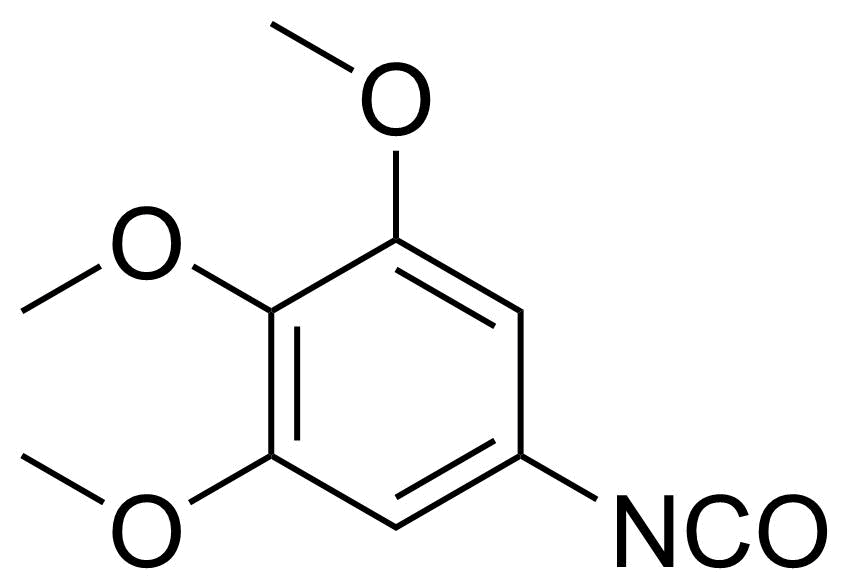 | [1016-19-9] | GEO-02411 | |
| 2,4,6-Trimethylphenyl isocyanate | 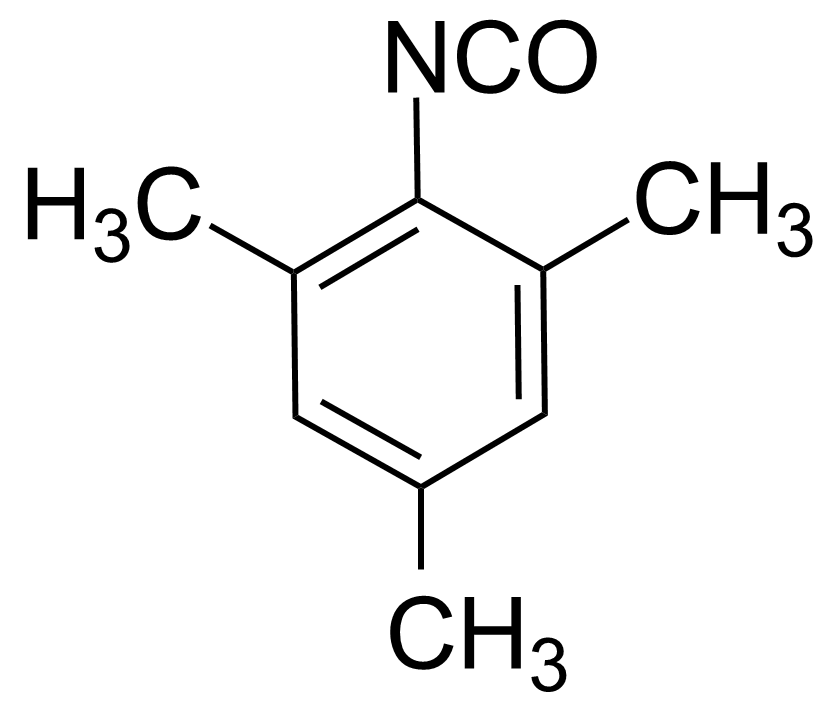 | [2958-62-5] | GEO-02422 | |
| Trimethylsilyl isocyanate | 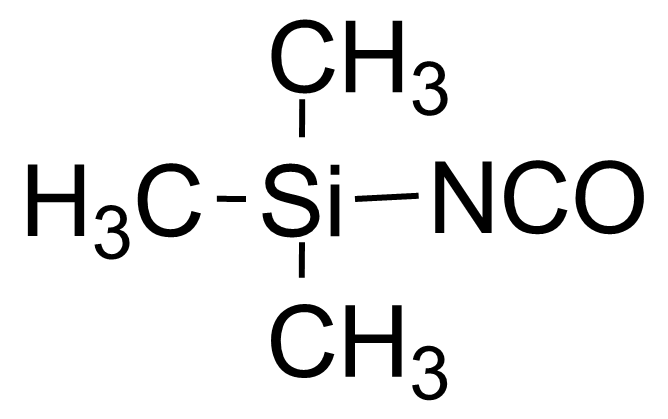 | [1118-02-1] | GEO-02424 |