Halides / Halide containing compounds
Organic halides are a large group of chemicals that contain at least one halogen atom, also known as halogenated compounds or organohalogens. They can be prepared by different type of halogenations (radical, electrophilic addition, α-keto halogenation) which depends on nature of substrate. The facility of halogenation is influenced by the halogen. Fluorine and chlorine are more electrophilic and are more aggressive halogenating agents while bromine is a weaker and iodine is the least reactive of them all. The facility of reactivity follows the reverse trend: iodine is most easily removed from organic compounds and organofluorine compounds are highly stable. Many organohalogens are important synthetic intermediates, suitable for further transformations like substitutions or couplings. They are often used in industry as solvents, pesticides, refrigerants, fire-resistant oils, ingredients of elastomers, adhesives and sealants, electrically insulating coatings, plasticizers, and plastics.
| Product name | Structure | CAS# | G-code | |
|---|---|---|---|---|
| New | 2-Bromo-7-methylquinoline-4-carboxylic acid | 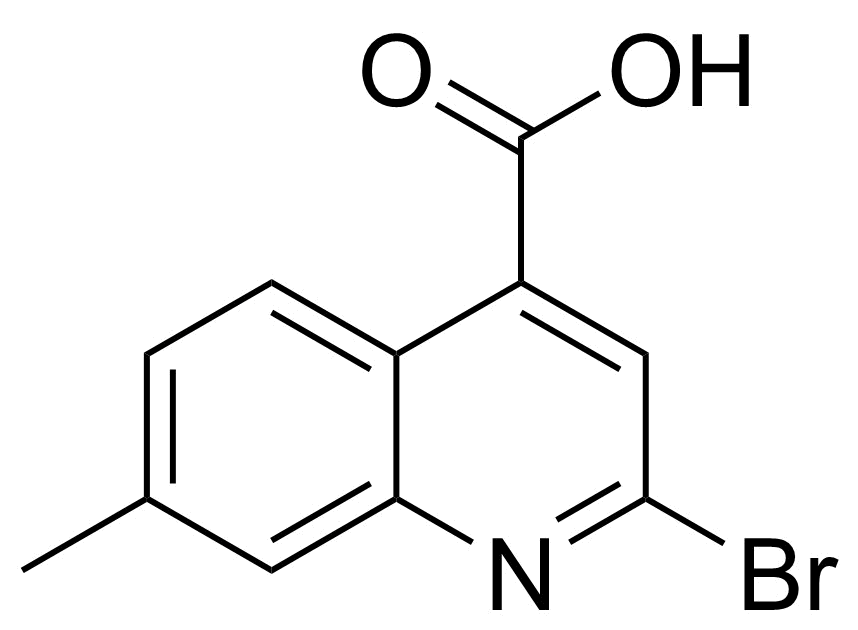 | [1267333-06-1] | GEO-04865 |
| 2-Bromo-4-methyl-1,3-thiazole-5-carboxylic acid | 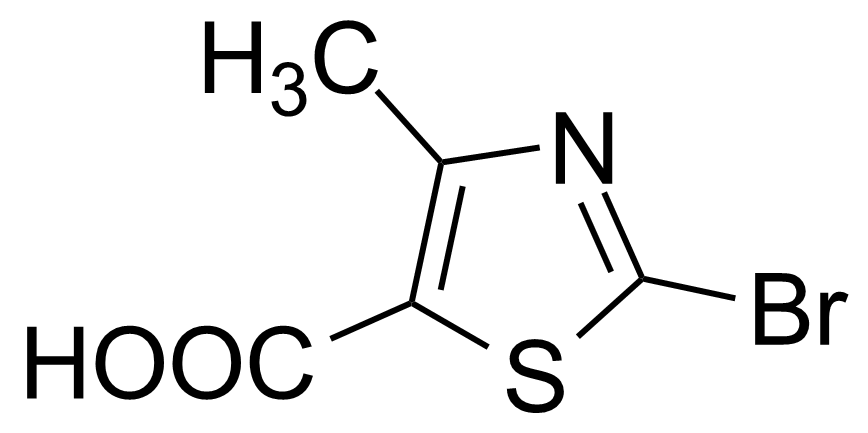 | [40003-41-6] | GEO-02509 | |
| 3-Bromo-4-methylthiophene | 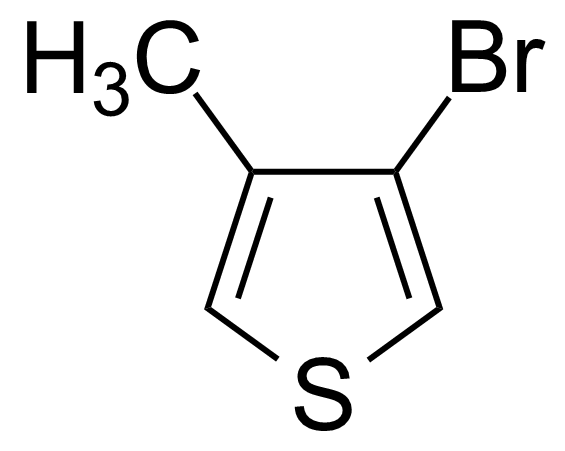 | [30318-99-1] | GEO-00513 | |
| 2-Bromo-5-methylthiophene | 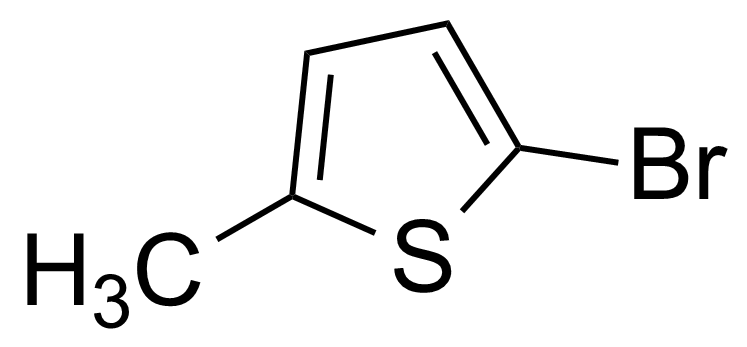 | [765-58-2] | GEO-00512 | |
| 3-(Bromomethyl)thiophene | 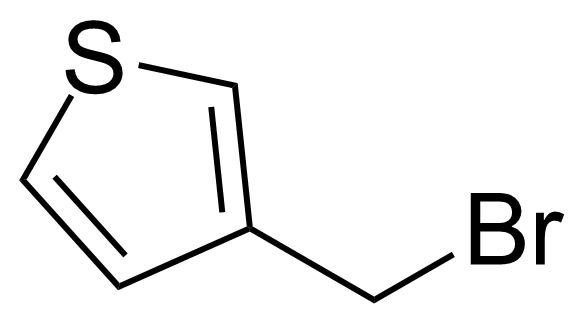 | [34846-44-1] | GEO-03030 | |
| 4-Bromo-2-methylthiophene | 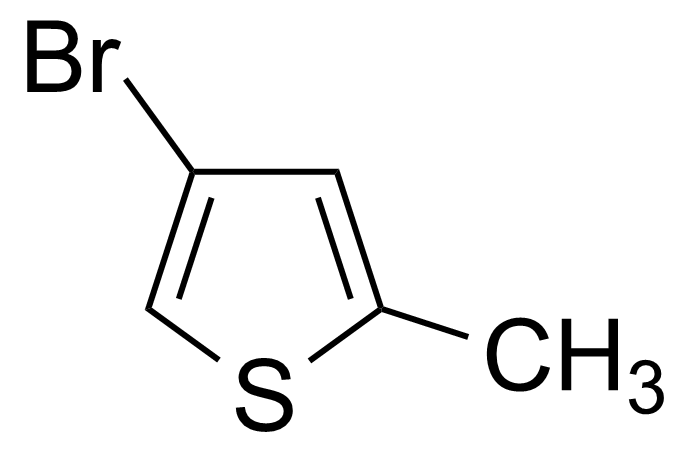 | [29421-92-9] | GEO-00511 | |
| 2-Bromo-3-methylthiophene | 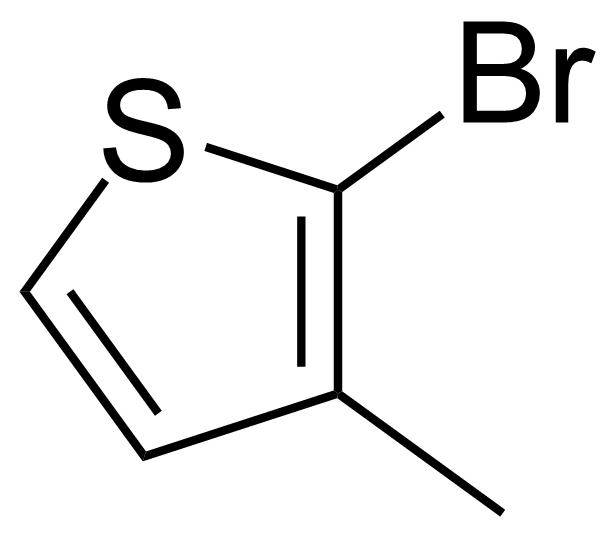 | [14282-76-9] | GEO-00510 | |
| 2-(Bromomethyl)thiophene | 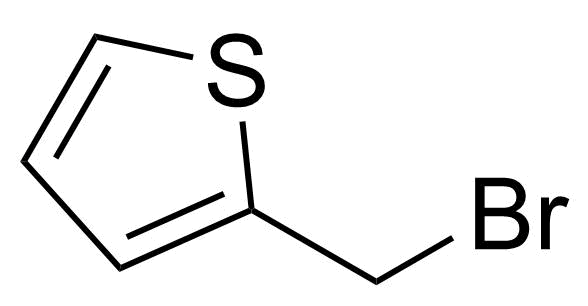 | [45438-73-1] | GEO-04883 | |
| 4-Bromo-5-methylthiophene-2-carboxaldehyde | 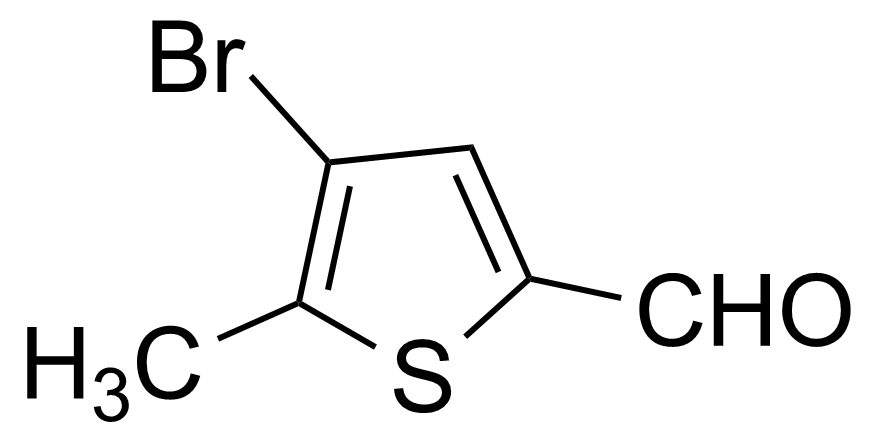 | [29421-75-8] | GEO-00516 | |
| 4-Bromo-3-methylthiophene-2-carboxaldehyde | 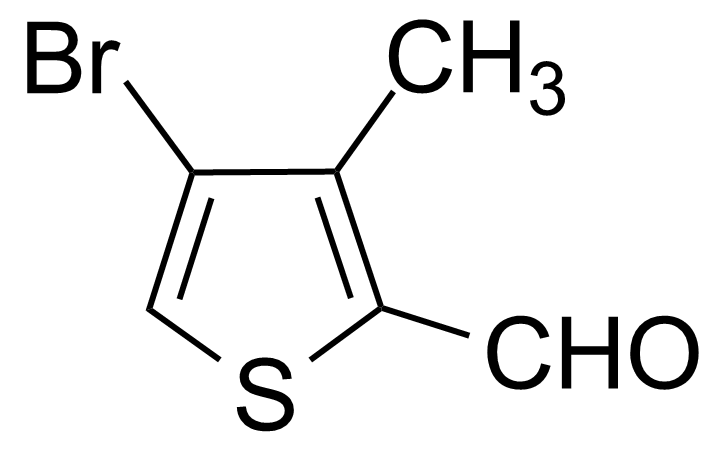 | [30153-47-0] | GEO-00515 | |
| 3-Bromo-5-methylthiophene-2-carboxaldehyde | 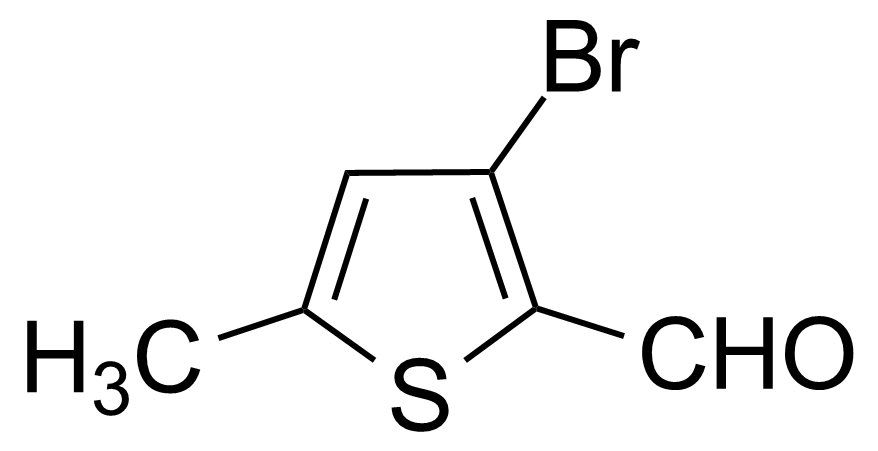 | [36155-82-5] | GEO-00514 | |
| 2-Bromo-6-nitrophenol | 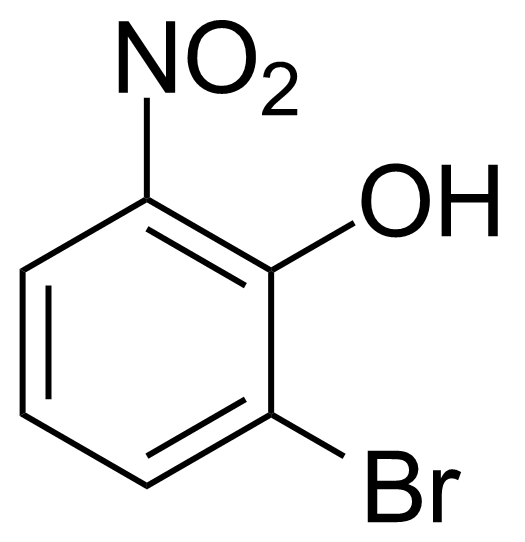 | [13073-25-1] | GEO-00526 | |
| 5-Bromo-3-nitropyridine-2-carbonitrile | 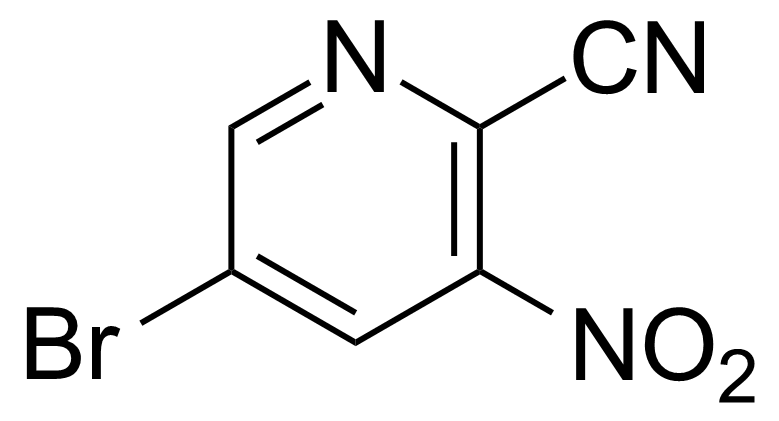 | [573675-25-9] | GEO-02510 | |
| 2-Bromo-5-nitrothiophene | 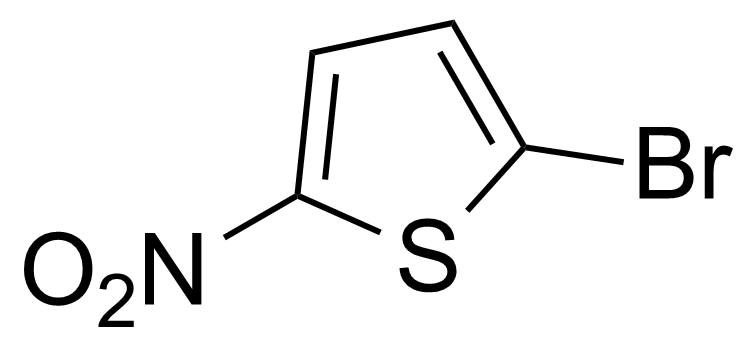 | [13195-50-1] | GEO-00528 | |
| 3-Bromo-2-nitrotoluene | 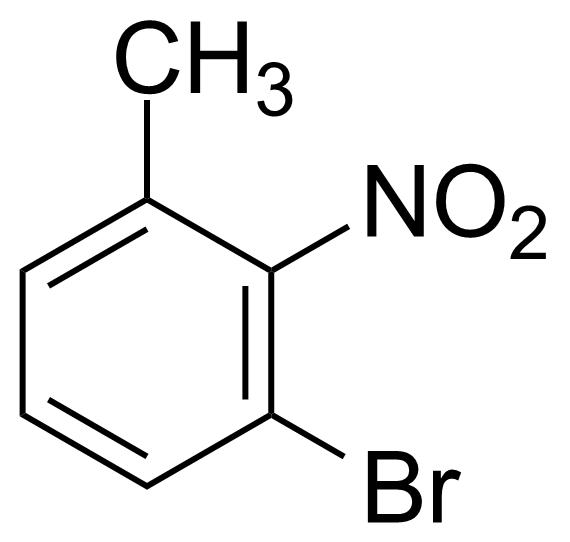 | [52414-97-8] | GEO-00529 | |
| 9-Bromo-1-nonene | 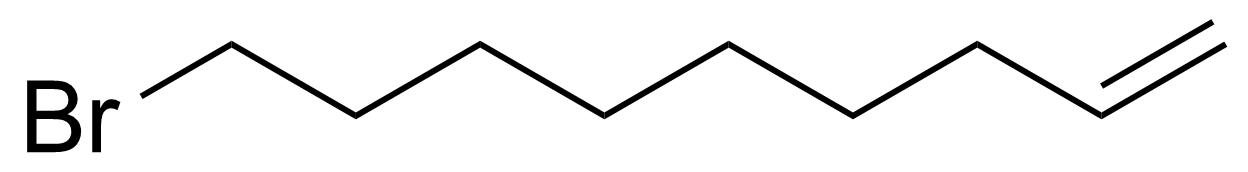 | [89359-54-6] | GEO-00531 | |
| 3-Bromooctane | 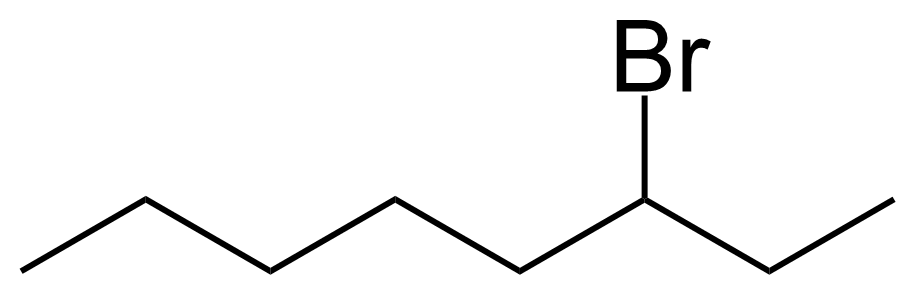 | [999-64-4] | GEO-04449 | |
| 8-Bromooctanol | 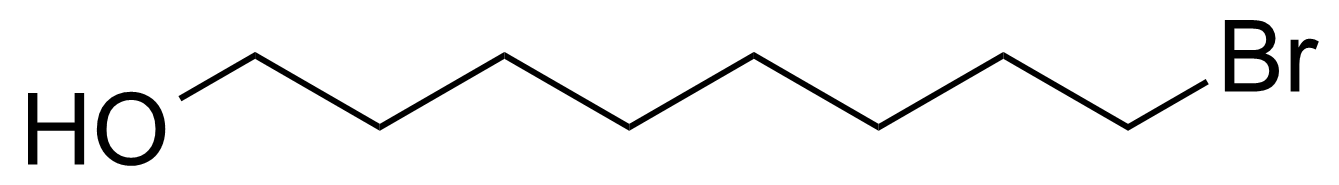 | [50816-19-8] | GEO-04211 | |
| 8-Bromo-1-octene | 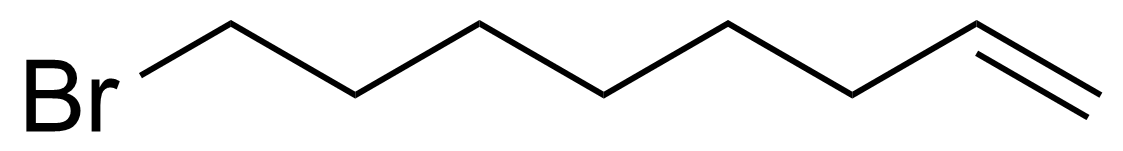 | [2695-48-9] | GEO-00534 | |
| 5-Bromooxindole | 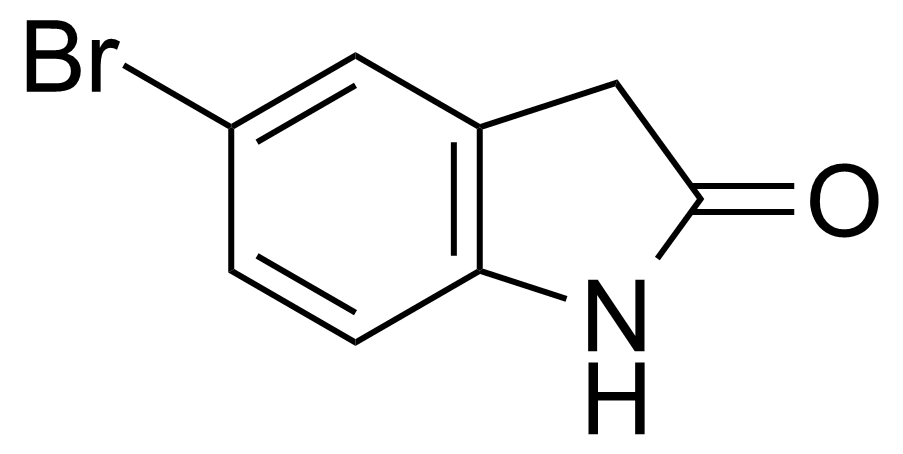 | [20870-78-4] | GEO-00536 |