Amines / Amine salts / Imines
Compounds that contain imino or amino functional group. Imines are prepared by condensation of aldehyde/ketone with appropriate amine. Imines are widely used as intermediates in the synthesis of heterocycles. Subsequent reduction of imine lead to amine. Amines are useful substrates in organic chemistry. Aside from their basicity, the dominant reactivity of amines is their nucleophilicity. They can undergo alkylation, acylation, sulfonation and diazotation reactions and many more. Most of primary amines are good ligands for metal ions to give coordination complexes. They are ubiquitous in biology as amino acids and neurotransmitters. They have a wide use in preparation of dyes and drugs (approx. 40% of drugs contain amine function group). Amines are often used as epoxy resin curing agents.
| Product name | Structure | CAS# | G-code | |
|---|---|---|---|---|
| Trimethylamine, 40% (w/w) Aqueous solution | 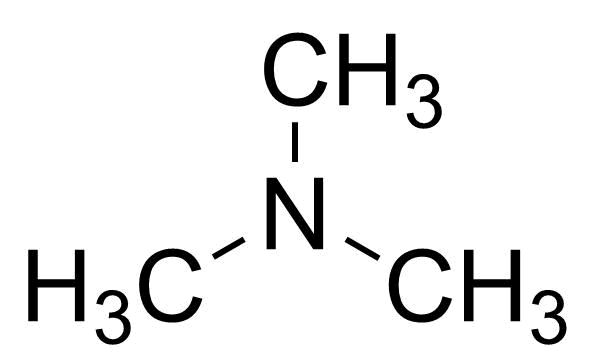 | [75-50-3] | GEO-04795 | |
| Trimethylamine, 33% (w/w) in Ethanol denatured with 2% Cyclohexane | 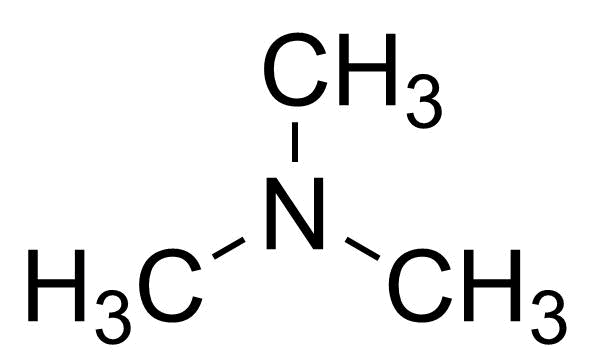 | [75-50-3] | GEO-04775 | |
| Trimethylamine, 1M in Tetrahydrofuran | 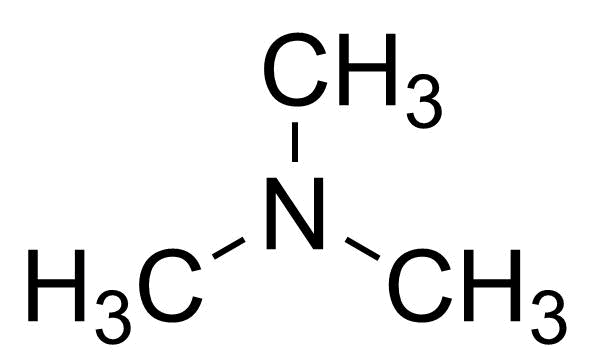 | [75-50-3] | GEO-04766 | |
| Trimethylamine, 33% (wt/wt) in Ethanol | 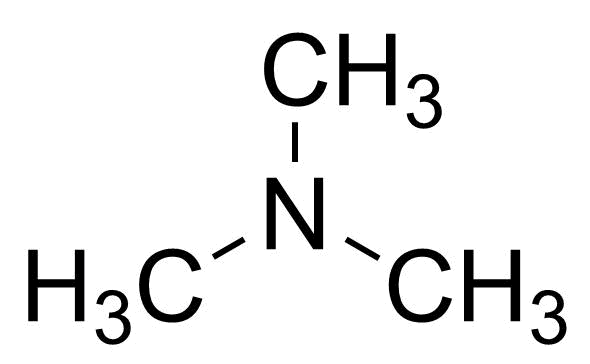 | [75-50-3] | GEO-04757 | |
| 2,7,7-Trimethyl-4-(5-methylfuran-2-yl)-5-oxo-1,4,5,6,7,8-hexahydroquinoline-3-carbonitrile |  | N/A | GEO-03599 | |
| 2,7,7-Trimethyl-4-(5-((5-methyl-1,3,4-thiadiazol-2-yl)thio)furan-2-yl)-5-oxo-1,4,5,6,7,8-hexahydroquinoline-3-carbonitrile |  | N/A | GEO-03524 | |
| N’,N’,N-Trimethyl-N-(2-(thiophen-2-yl)quinolin-4-yl)ethane-1,2-diamine hydrobromide | 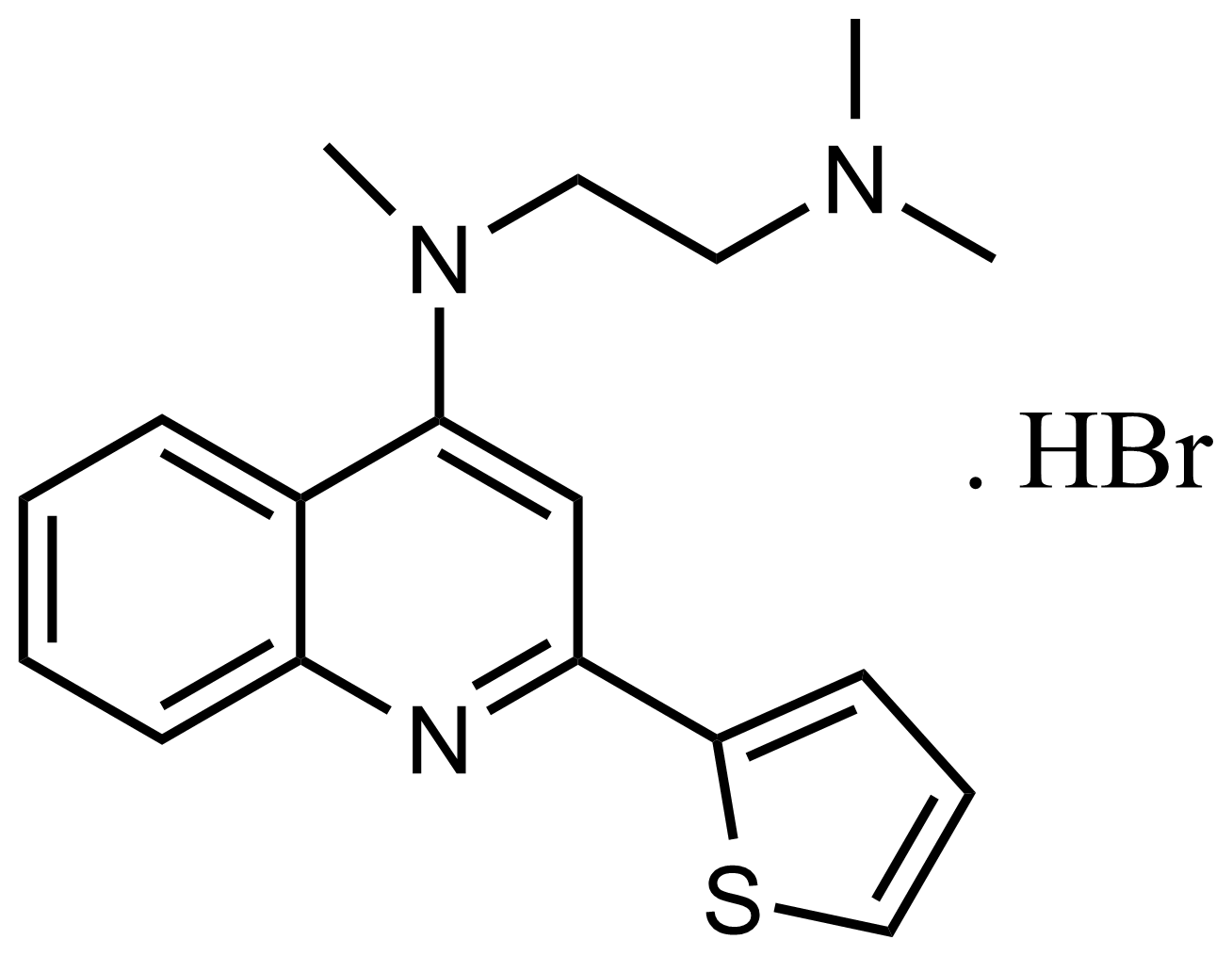 | N/A | GEO-03351 | |
| Tris(2-dimethylaminoethyl)amine | 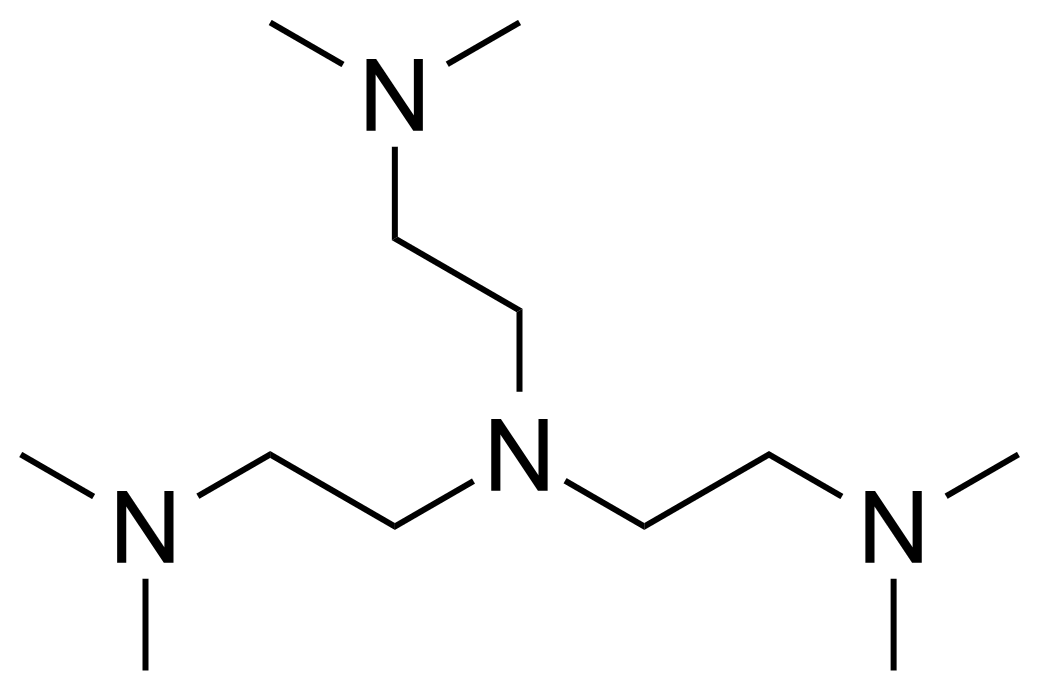 | [33527-91-2] | GEO-04438 | |
| Tris(4-formylphenyl)amine | 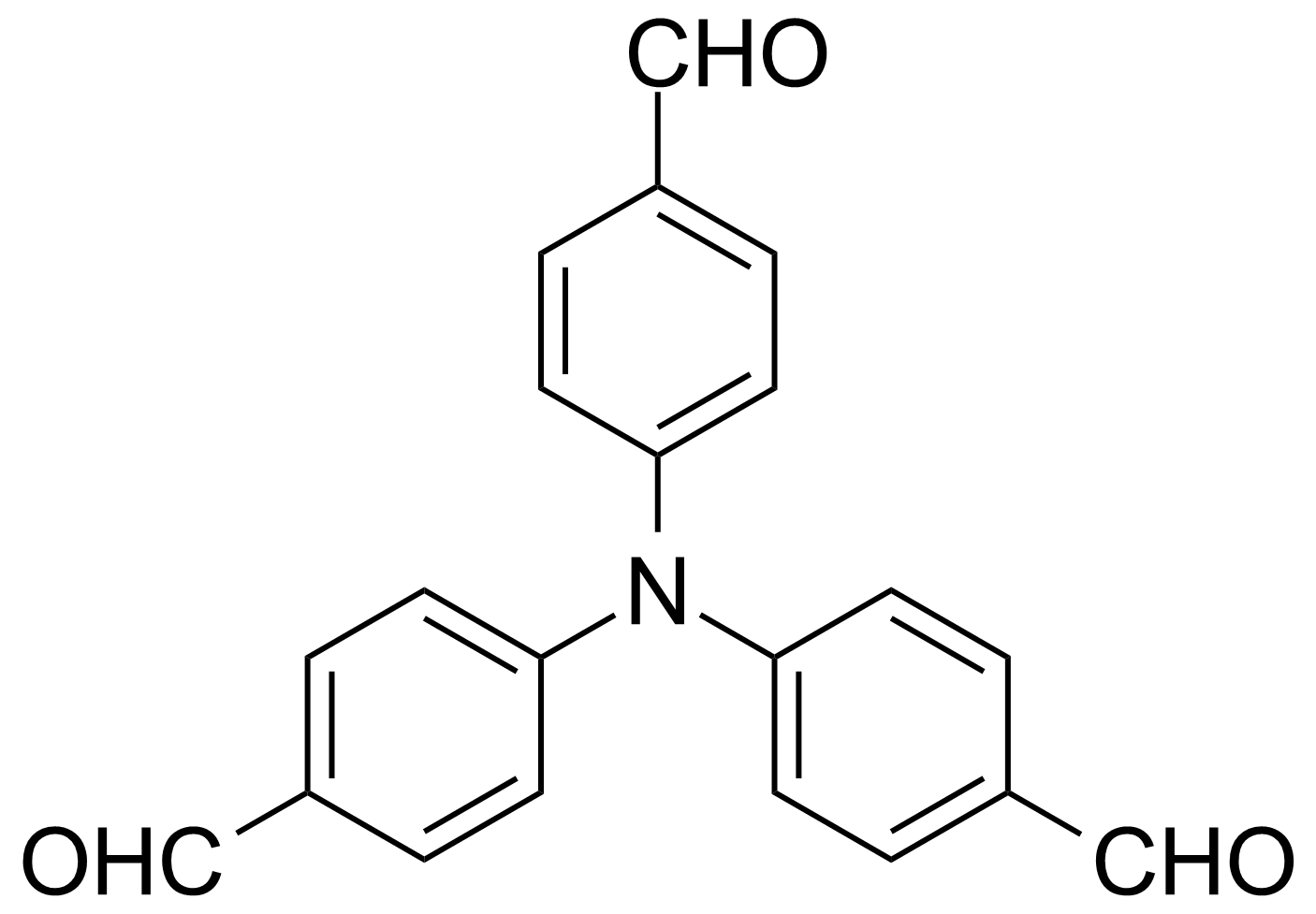 | [119001-43-3] | GEO-02432 | |
| New | Vorinostat | 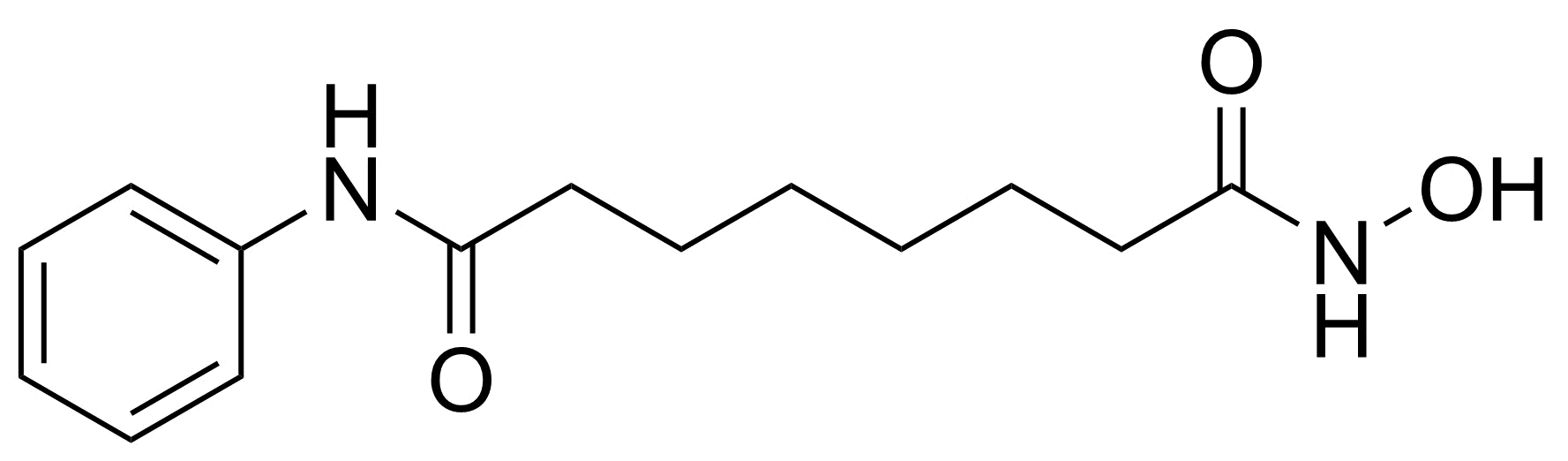 | [149647-78-9] | GEO-04933 |