In stock
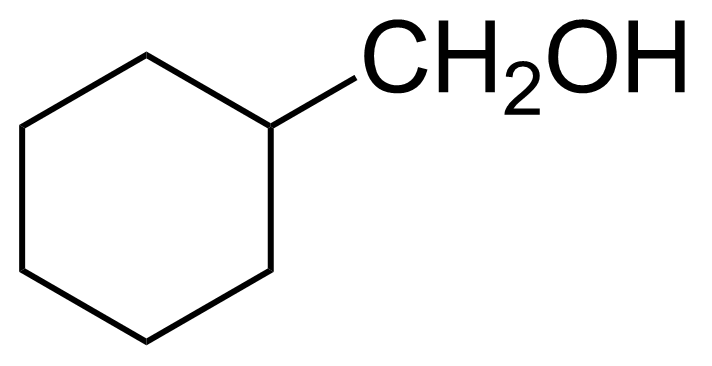
Cyclohexanemethanol
Product has been discontinued; however, we still have inventory in stock.
CAS#[100-49-2]
G-codeGEO-00862
EC number202-857-8
Molecular formulaC7H14O
Molecular weight114.19
Synonyms
(Hydroxymethyl)cyclohexane ; Hexahydrobenzyl alcohol ; Cyclohexylcarbinol ; Cyclohexylmethanol
For more information or to place an inquiry, please email us to
georganics@georganics.sk or use our contact form
Regulatory Information
Not a hazardous substance or mixture according to Regulation (EC) No. 1272/2008.
Product categorization
Main category
Second level
Description
Cyclohexanemethanol is usually used as a reagent in organic synthesis. It acts as a nucleohpile in highly beta-selective glucosidation of…
Show full descriptionGeneral description of Cyclohexanemethanol:
Cyclohexanemethanol (CM), or (hydroxymethyl)cyclohexane, or cyclohexylcarbinol [100-49-2] is six-membered alicyclic organic compound that belong to alcohols. In its pure form, it is colorless viscious highly flammable liquid with the boiling point of 179-180 °C.[1] It is insoluble in water and soluble in common organic solvents. CM is a non-corrosive and quick volatilize liquid with the LD50 = 250 mg/kg (mouse, intraperitoneal).[2] Convenient laboratory preparation starts with chlorocyclohexane that is converted to cyclohexylmagnesium chloride, which reacts with formaldehyde or paraformaldehyde.[3]Application of Cyclohexanemethanol:
Cyclohexanemethanol is usually used as a reagent in organic synthesis. It acts as a nucleohpile in highly beta-selective glucosidation of ethylthioglucosides.[4] It can be used as a starting material for the synthesis of cyclohexanecarboxaldehyde, cyclohexanecarboxylic acid, cyclohexanone, and 1,4-cyclohexadione by photocatalytic oxidation using titanium dioxide nanoparticles.[5] Cyclohexanemethanol can be biologically oxidized by the yeasts Candida maltosa and Trichosporon mucoides under laboratory conditions.[6]Important Notes:
Store in cool place. Keep container tightly closed in a dry and well-ventilated place. Incompatible materials are Strong oxidizing agents, Acid chlorides, Acid anhydrides. Excess heat. Keep away from open flames, hot surfaces and sources of ignition. Hazardous Combustion Products: Carbon monoxide (CO), Carbon dioxide (CO2). Cyclohexanemethanol is stable under normal conditionsProduct categorization (Chemical groups):
Main category: Second level: ______________________________________________________________________________________ [1] H. Adkins, H. I. Cramer J. Am. Chem. Soc. 1930, 52, 4349. [2] https://chem.nlm.nih.gov/chemidplus/sid/0000100492. No author, National Technical Information Service. Vol. AD277-689 [3] H. Gilman, W. E. Catlin Org. Synth. 1926, 6, 22. [4] Y. Okada, T. Mukae, K. Okajima, M. Taira, M. Fujita, H. Yamada Org. Lett. 2007, 9, 1573. [5] O. S. Mohamed, S. A. Ahmed, M. F. Mostafa, A. M. A. Abdel-Wahab J. Photochem. Photobiol. A 2008, 209. [6] R. Schlüter, A. Dallinger, J. Kabisch, I. Duldhardt, F. Schauer Appl. Microbiol. Biotechnol. 2019, 103, 4137.
Similar products
| Product name | Structure | CAS# | G-code | |
|---|---|---|---|---|
| 1-Acenaphthenol | 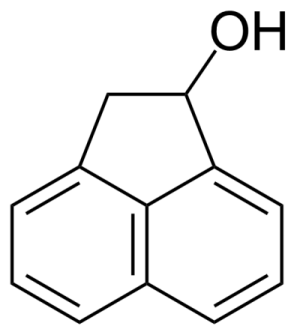 | [6306-07-6] | GEO-00001 | |
| 7-Acetamido-4-hydroxy-naphthalene-2-sulfonic acid | 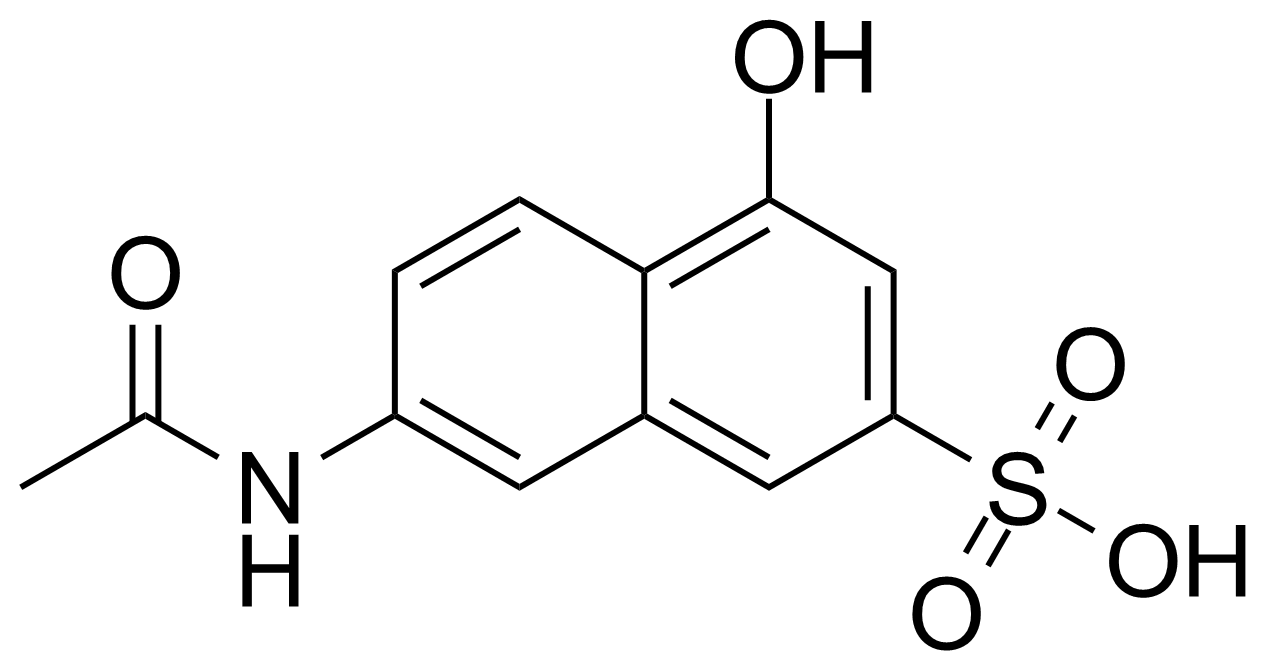 | [6334-97-0] | GEO-04013 | |
| New | 1,3-Adamantanediol | 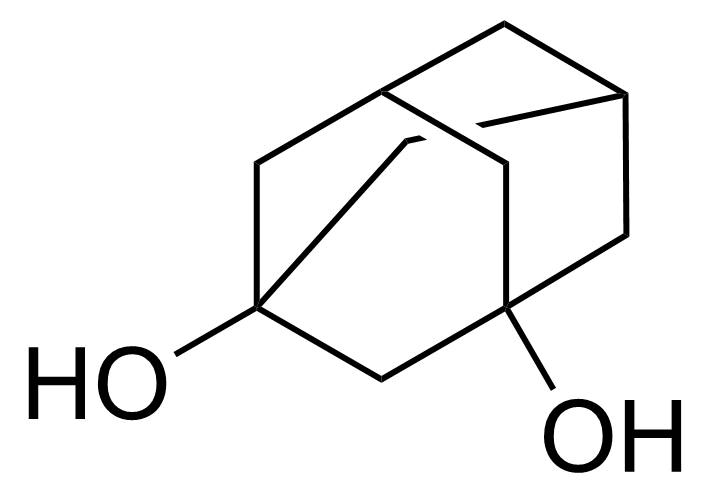 | [5001-18-3] | GEO-04858 |
| 1-Adamantanemethanol | 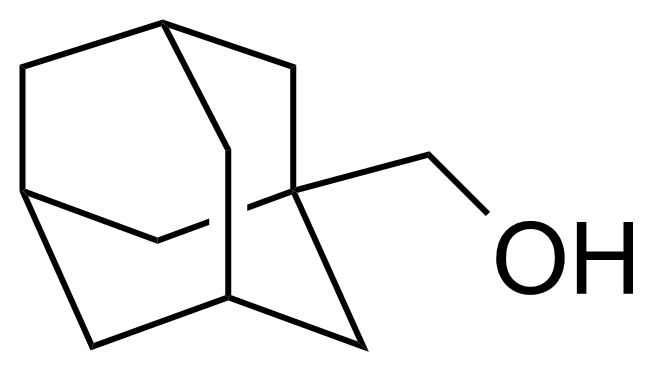 | [770-71-8] | GEO-04333 | |
| beta-D-Allopyranose | 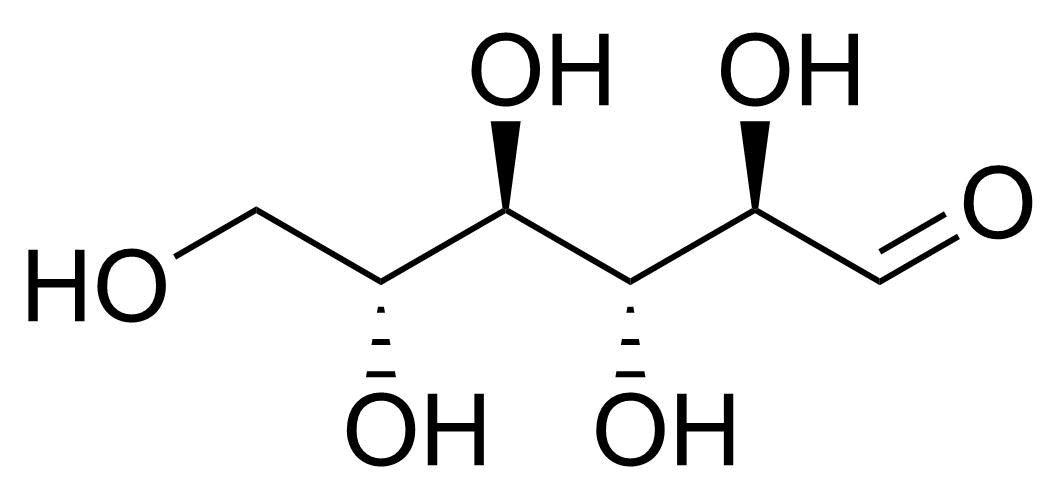 | [7283-09-2] | GEO-04660 | |
| D-Allose | 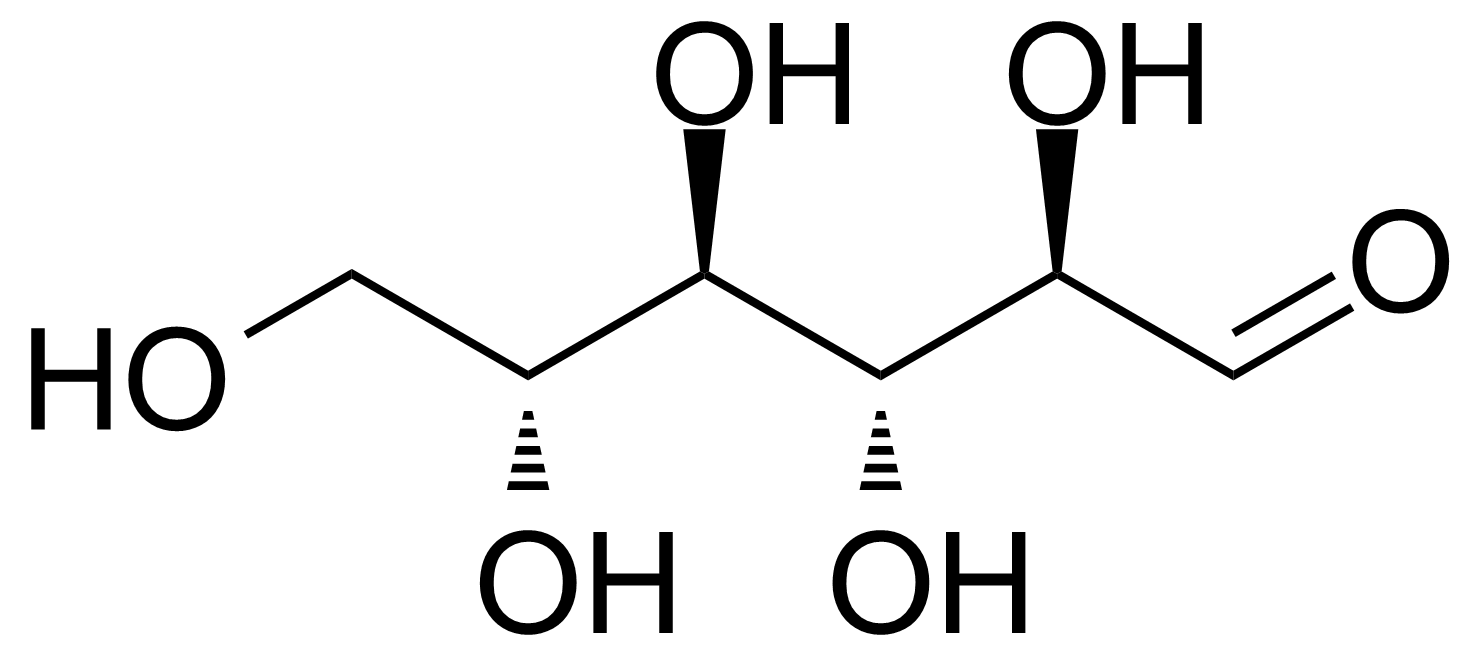 | [2595-97-3] | GEO-00057 | |
| L-Allose | 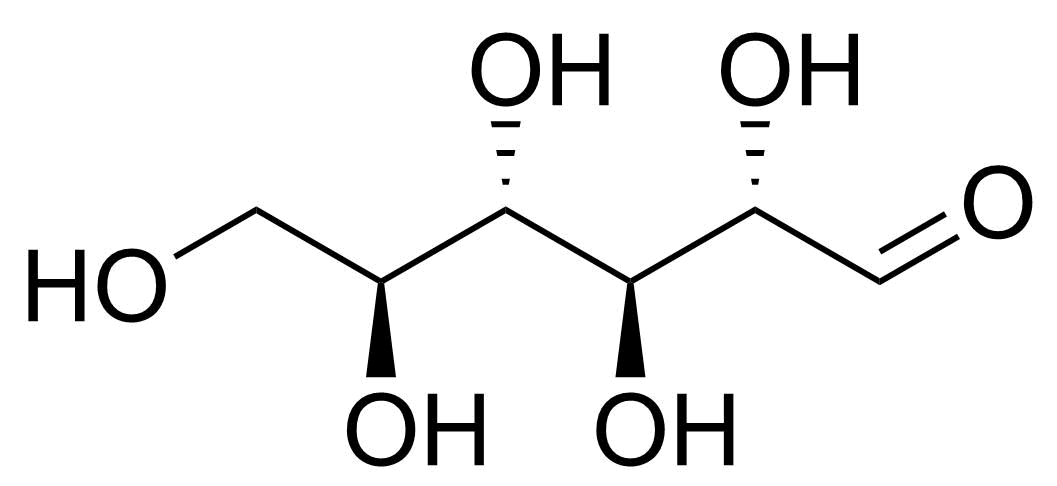 | [39392-62-6] | GEO-04661 | |
| 2-(Allyloxy)phenol | 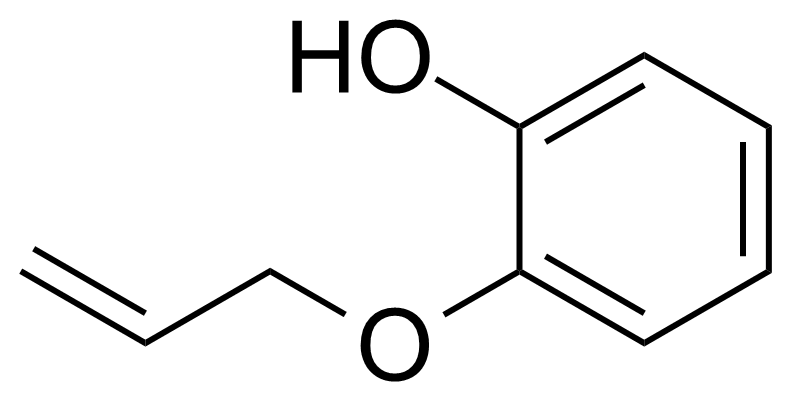 | [1126-20-1] | GEO-04471 | |
| D-Altrose | 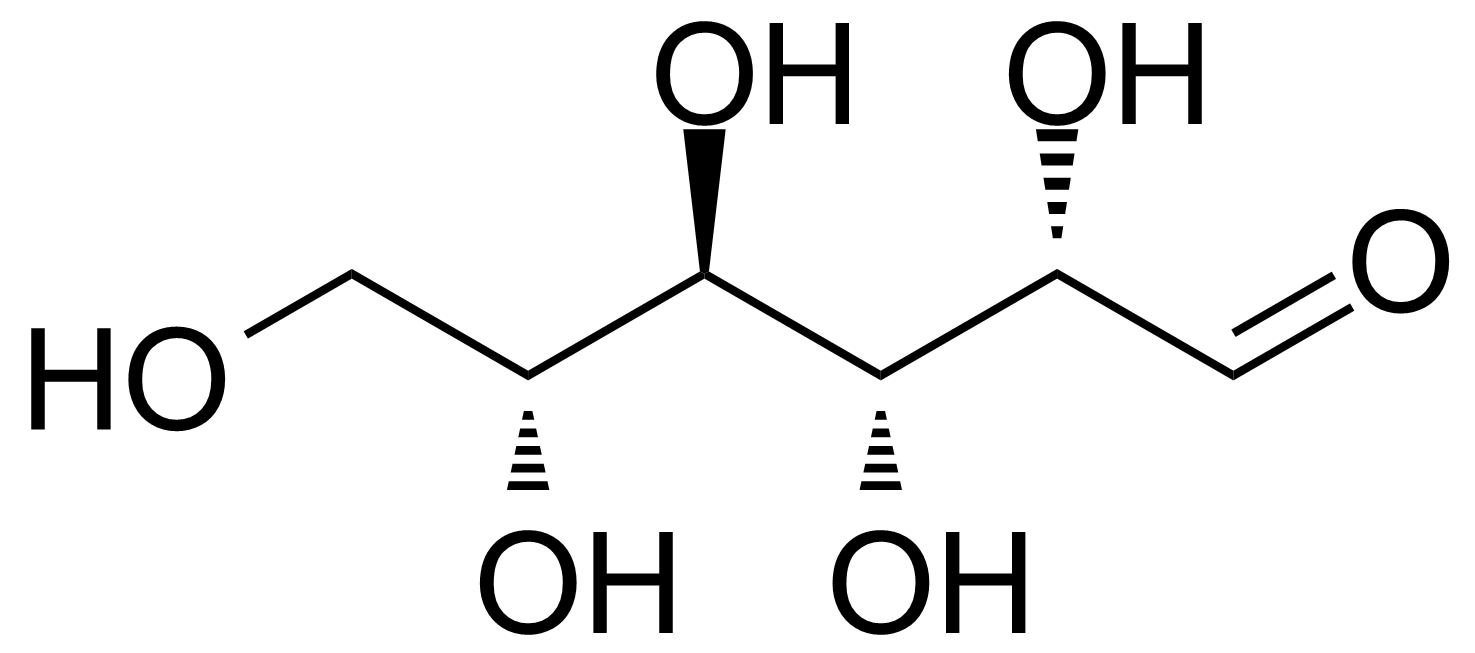 | [1990-29-0] | GEO-00058 | |
| L-Altrose | 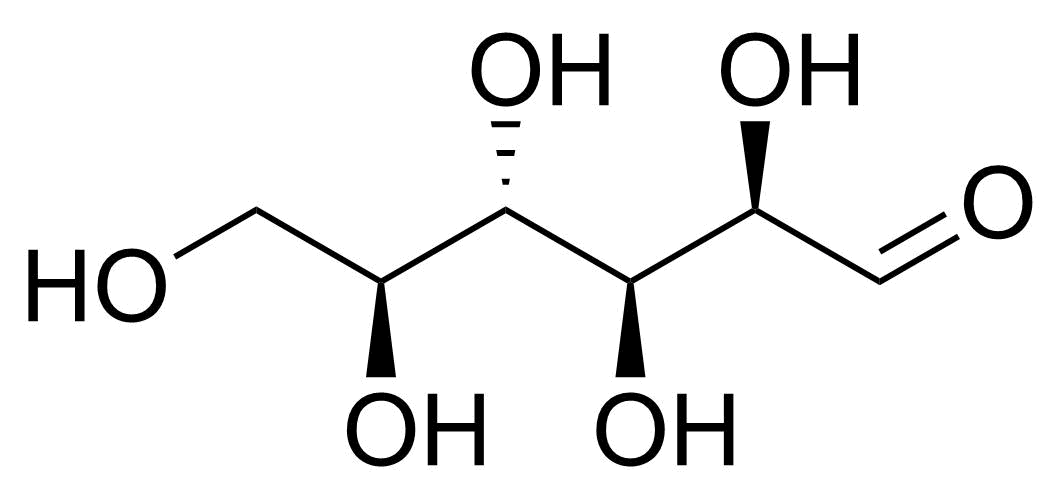 | [1949-88-8] | GEO-04662 |